Delayed radiation-induced inflammation accompanying a marked carbohydrate antigen 19-9 elevation in a patient with resected pancreatic cancer
Article information
Abstract
Although carbohydrate antigen (CA) 19-9 is a useful tumor marker for pancreatic cancer, it can also become elevated from a variety of benign and malignant conditions. Herein we describe an unusual presentation of elevated CA 19-9 in an asymptomatic patient who had previously undergone adjuvant chemotherapy and radiation therapy for resected early stage pancreatic cancer. The rise in CA 19-9 might be due to delayed radiation-induced inflammation related to previous intra-abdominal radiation therapy with or without radiation recall induced by gemcitabine. After treatment with corticosteroids the CA 19-9 level decreased to normal, and the patient has not developed any evidence of recurrent cancer to date.
Introduction
The modified Lewis blood group antigen, carbohydrate antigen (CA) 19-9, is a useful tumor marker for many patients with pancreatic cancer. The serum concentration of CA 19-9 is part of the standard workup for patients with newly diagnosed pancreatic cancer, and a cutoff value of 37 U/mL is associated with sensitivity for malignancy ranging from 70%—92% and specificity ranging from 68%—92% [1]. False positives may arise from a variety of benign conditions such as cirrhosis, cholangitis, cholestasis, and pancreatitis [1-3]. In patients who undergo curative-intent surgical resection of their tumor, the postoperative CA 19-9 level has also been shown to be an important prognostic factor, and is frequently obtained during the follow-up period to monitor for recurrence. We present the case of a woman with pancreatic cancer who underwent surgical resection followed by adjuvant chemotherapy and radiation therapy, who experienced an asymptomatic rise in CA 19-9 nine months after completing all adjuvant therapy that was thought to be inflammatory rather than malignant in nature. After treatment with steroids the CA 19-9 level decreased to normal and she currently has no evidence of disease.
Case Report
A 67-year-old female with a past medical history of smoking, emphysema, pulmonary hypertension, and hepatic steatosis presented with shortness of breath. She underwent a computed tomography (CT) scan of the chest, which was negative for any new pulmonary abnormalities, but did report an incidental finding of dilation of the main pancreatic duct. She denied any prior history of pancreatitis, nausea, vomiting, abdominal pain, back pain, jaundice or clay colored stools. A CT of the abdomen revealed a 12 mm × 8 mm hypodense mass in the uncinate process of the pancreas causing main pancreatic duct dilation and obstruction. Upper endoscopic ultrasound with fine needle aspiration of the mass showed atypical cells suspicious for malignancy. Initial CA 19-9 level was 50 U/mL. Metastatic workup was negative.
The patient underwent a Whipple’s pancreaticoduodenectomy in May 2013. Pathology showed a 1.7 cm low grade pancreatic ductal adenocarcinoma with no evidence of extra-pancreatic, lymphovascular or perineural invasion and 11 negative lymph nodes. The resection margins were negative, though carcinoma-in-situ extended up to 2 mm from the retroperitoneal margin. The American Joint Committee on Cancer (AJCC) pathologic stage was pT1N0 (stage IA). Her postoperative course was complicated by Clostridium difficile colitis treated with metronidazole and vancomycin. Adjuvant therapy included 1 cycle intravenous gemcitabine 1,000 mg/m2 from July 3 through July 17, 2013, followed by radiation therapy to the tumor bed with concurrent infusional 5-FU 250 mg/m2/day from August 12 through September 19, 2013, followed by 3 additional cycles of gemcitabine 1,000 mg/m2 from October 15—December 10, 2013.
Intensity-modulated radiation therapy (IMRT) was used to deliver 45 Gy to the pancreatic bed and regional lymph nodes followed by an additional 5.4 Gy boost to the tumor bed, to a total dose of 50.4 Gy. All planning constraints for organs-at-risk were met [4]. The mean liver dose was 19.8 Gy, and the volume of liver receiving at least 30 Gy was 27.2%. The maximum dose to bowel was 54.8 Gy and the volume of bowel receiving at least 45 Gy was 194 mL. A representative axial slice of the treatment plan is shown in Fig. 1A. She tolerated the treatment without any acute side effects. She had no evidence of recurrence through April 2014 when she underwent ventral incisional hernia repair (including some lysis of adhesions) with gore-tex dual mesh.
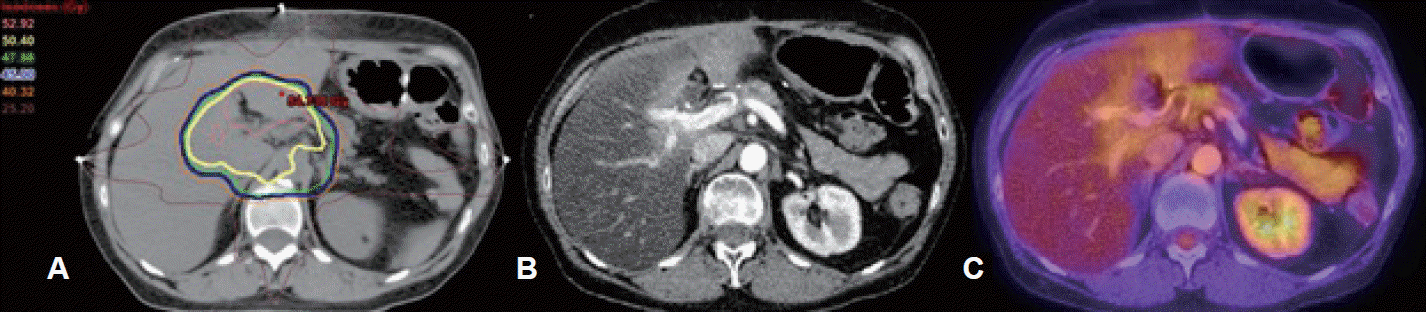
(A) Representative axial slice from intensity modulated radiation therapy treatment plan, (B) follow-up computed tomography (CT), and (C) positron emission tomography (PET)/CT scans 12 months after the completion of radiotherapy at the time of the elevation in carbohydrate antigen (CA) 19-9, respectively.
In September 2014, approximately 9 months after completion of gemcitabine and 12 months after completion of radiotherapy, CA 19-9 rose for the first time to 287 U/mL. Repeat measurement in October 2014 was 2,885 U/mL. This was associated with a grade 1 transaminase elevation according to the common terminology criteria for adverse events version 4.0 without an increase in bilirubin or alkaline phosphatase [5]. Kidney function was normal. The patient remained completely asymptomatic. Positron emission tomography (PET)-CT at this time showed increased attenuation, enhancement and hypermetabolic activity in the medial aspect of the liver, which corresponded closely to the high dose region of the radiation therapy plan without recurrence (Fig. 1). Her case was discussed at multidisciplinary tumor board, and it was thought that the elevation in CA 19-9 was more likely due to a radiation-induced inflammatory or fibrotic reaction than tumor recurrence, and as such she was started on prednisone 60 mg daily.
Within 1 month CA 19-9 decreased to 248 U/mL, and by 3 months it returned to baseline with resolution of the transaminase elevation as well. As the CA 19-9 decreased she was gradually tapered off of the prednisone, with the last dose being in January 2015. The CA 19-9 continues to be at baseline and she has had no evidence of recurrence after 3 years of follow-up.
Discussion
A variety of benign and malignant conditions can lead to elevations in CA 19-9 [1-3]. Though most commonly utilized as a tumor marker in pancreatic cancer, it can also be elevated by several other intra-abdominal malignancies. The most common nonmalignant causes include either obstructive jaundice, due to the hepatic clearance of CA 19-9 via the bile, or to inflammatory hypersecretion of CA 19-9 by epithelial cells (biliary or otherwise) [6]. Inflammatory processes that have been associated with elevated CA 19-9 include chronic liver disease, pancreatitis, collagen vascular disease, diabetes mellitus, lung disease, thyroid disease, and excessive tea consumption [7-11]. Based on the complete response of our patient’s CA 19-9 level to corticosteroids, and lack of pancreatic cancer recurrence with longer follow-up, it is likely that the observed abnormality was due to inflammation rather than malignancy. The presence of metabolic activity in the irradiated liver on PET/CT 12 months after the completion of radiation therapy, with a concurrent grade 1 transaminase elevation that mirrored the rise and fall of CA 19-9, implicates an active radiation-related inflammatory process in the liver as the source of the CA 19-9 abnormality. This process could represent delayed radiation-induced inflammation with or without a radiation recall reaction related with gemcitabine. Either way, in this case presentation we have described, to our knowledge for the first time, an example of marked CA 19-9 elevation due to delayed inflammation related to intra-abdominal radiation therapy.
Radiation recall is an uncommon event that can be defined as inflammation that develops in a previously irradiated area due to subsequent exposure to an inciting agent, most commonly cytotoxic chemotherapy [12,13]. Radiation recall most commonly involves the skin, and occurs within days to weeks after the first dose of an inciting agent [12,13]. Some of these reactions may occur up to 15 years from the radiotherapy. Gemcitabine has been reported to preferentially cause radiation recall in visceral organs and there is more likely to be a longer lag time between the administration of the causative agent and the appearance of symptoms [14,15]. If our patient did experience radiation recall due to gemcitabine, one unusual aspect of the case would be for the recall reaction to take place as long as 9 months after completion of the gemcitabine. Some previously reported gemcitabine-induced recall reactions have occurred within weeks of discontinuing gemcitabine [16,17], but none have been reported to take place months later. Of note, the patient denied taking any other supplements prior to this episode as well. This would also be the first reported case of radiation recall in an asymptomatic patient whose primary abnormality from the recall reaction is a tumor marker elevation.
Other potential causes of elevated CA 19-9 seem less plausible. For instance, the bilirubin never became abnormal, suggesting against any problem with hepatic metabolism or excretion of bile. Excessive alcohol use has been implicated in raising CA 19-9 in the context of extreme pathologic cholestasis, and can increase acute and late radiation toxicity via S-adenosylmethionine and glutathione depletion in the liver [18], but it is unlikely that our patient’s light alcohol consumption (i.e., occasional single glass of wine with dinner) contributed significantly in this case. The syndrome of radiation induced liver disease is not suggested by the clinical picture, as it usually develops between 2 weeks and 4 months after RT, and is characterized by hepatomegaly, ascites, and increased liver function tests (particularly alkaline phosphatase) [19]. Our patient did have a distant history of smoking and active lung disease, but her intermittent pulmonary exacerbations did not lead to CA 19-9 elevation on other occasions and on imaging her lungs demonstrated no evidence of worsened inflammation or pneumonia during this episode. Thyroid disease can raise CA 19-9 but our patient’s thyroid stimulating hormone levels were within normal limits in both March 2013 and March 2015, and she has no prior history of thyroid disease.
According to the 2006 American Society of Clinical Oncology (ASCO) expert panel, rising CA 19-9 levels after surgery usually precede the radiographic appearance of recurrent disease, but confirmation of disease progression should be pursued with imaging studies and/or biopsy [20]. This case presentation highlights the importance of this statement in patient care, as initiating chemotherapy for the elevation in CA 19-9 alone would have resulted in inappropriate treatment and unwarranted side effects. We suggest that elevations in CA 19-9 with other evidence of delayed radiation-induced inflammation should prompt clinicians to consider the radiation or radiation recall as the cause of the elevated tumor marker, and treat it accordingly with careful monitoring to ensure an appropriate decrease in the CA 19-9 and that no underlying cancer recurrence does develop thereafter.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.