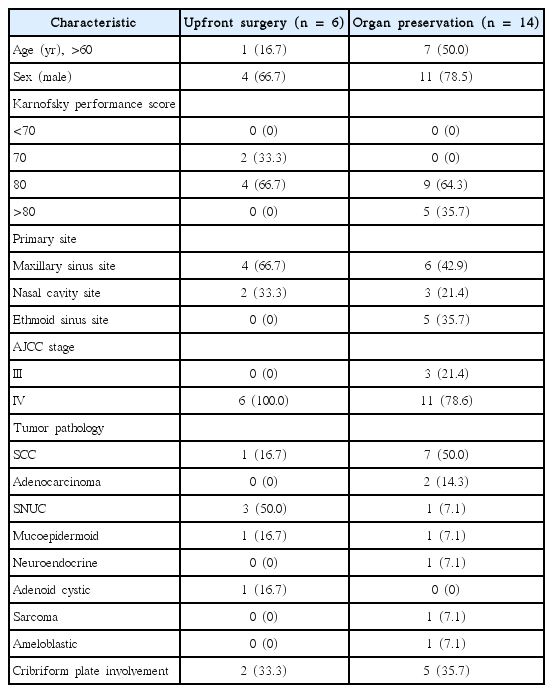
Organ preservation with neoadjuvant chemoradiation in patients with orbit invasive sinonasal cancer otherwise requiring exenteration
Article information
Abstract
Purpose
We sought to determine if organ preservation (OP) with neoadjuvant chemoradiation (CRT) was feasible in patients with sinonasal cancer determined to require exenteration.
Materials and Methods
Twenty patients were determined to require exenteration for definitive treatment from 2005 to 2014. Fourteen patients underwent OP and 6 patients received exenteration with adjuvant CRT. Exenteration free survival (EFS), locoregional control (LRC), progression-free survival (PFS), and overall survival (OS) were estimated.
Results
Five patients (36%) receiving OP had complete disease response at time of surgery. With a median follow-up of 18.8 months, EFS was 62% at 2 years for patients undergoing OP. At 2 years, there were no significant differences in LRC, PFS or OS (all all p > 0.050) between the groups. Less grade 3 or greater toxicity was seen in patients undergoing OP (p = 0.003). Visual function was preserved in all patients undergoing OP.
Conclusion
For patients with sinonasal cancer, OP may avoid exenteration, offering similar disease control and improved toxicity.
Introduction
Sinonasal cancer has an estimated incidence of 0.556 cases per 100,000 people in the United States, comprising up to 5% of all upper aerodigestive tract cancers [1]. Early stage disease often has minimal symptoms, which when combined with the compact anatomy of the paranasal sinuses result in a significant proportion of sinonasal cancers presenting at locally advanced stages [2]. While multiple published studies indicate the maxillary sinus as the most common primary site of tumor origin, the advanced stage at which many of these cancers present can further obscure the true site of origin [1,3-5]. Additionally, locally advanced sinonasal cancer can encroach upon the orbital contents and is a prognostic factor influencing survival [6].
The incidence of orbital invasion varies with site of origin and histology among sinonasal cancers, with squamous cell carcinoma (SCC) of the maxillary antrum invading the orbit in up to 80% of cases [7]. In patients with cancers involving the soft tissue of the orbit, orbital exenteration has been shown to result in similar outcomes as patients with bone only involvement of the orbit not receiving an exenteration as part of their surgical therapy [7], and has become the standard treatment at many centers for patients with soft tissue involvement of the orbit. Even following orbital exenteration, a significant proportion of patients with orbit invasive sinonasal cancer require an aggressive adjuvant treatment regimen composed of additional radical surgical resection and postoperative radiotherapy [8]. Patients receiving these regimens can be left with significant disfigurement and impaired functional problems. Even with combined modality therapy, local failure remains a common site of recurrence [8].
Given the subsequent challenges in functional status and quality of life impact of an orbital exenteration, alternative approaches have been tried in lieu of surgical resection and combined with different surgical techniques [8-15]. However, the impact of a strategy of organ preservation (OP) on oncologic outcomes is not fully known [12]. It has been shown in surgically unresectable patients that single modality therapy using either radiation or chemotherapy alone is suboptimal [16]. Therefore the possibility of multimodality therapy with the intention of OP is greatly appealing, with an ideal regimen balancing local control and survival rates with the preservation of ocular function.
In this study, we examined patients from a single institution with orbit invasive sinonasal cancer. We sought to compare survival and disease control in patients receiving neoadjuvant chemoradiation (CRT) in an OP approach with patients receiving upfront orbital exenteration followed by adjuvant therapy.
Materials and Methods
As part of University of Louisville Institutional Review Board approved study, which was granted a waiver of informed consent, we reviewed our prospective database of all patients who presented to our multidisciplinary head and neck cancer clinic from 2005 to 2014. Over this time period, our institutional practice has shifted from definitive surgery with orbital exenteration to OP if deemed appropriate by the multidisciplinary clinical members.
1. Study population and data
All patients underwent a full staging workup including diagnostic neck imaging, chest imaging, and evaluation by a head and neck surgeon, radiation oncologist, and medical oncologist. We included patients who had a biopsy proven malignancy arising from the nasal cavity, maxillary, or ethmoid sinus and had evidence of orbital invasion that would require orbital exenteration as part of definitive surgical intervention (as determined by a multidisciplinary tumor board). Patients were excluded who were determined to have distant metastases at time of original staging workup, previous history of head and neck cancer, synchronous cancer diagnosis, or who did not receive definitive treatment at our institution.
Demographic information including age, sex, and ethnicity were collected. American Joint Committee on Cancer (AJCC) T-stage and N-stage were determined at time of initial presentation by available clinical, pathologic, and radiographic data. Staging was updated at time of analysis in accordance with the seventh edition of the AJCC staging. Extent of orbital encroachment was determined by computed tomography (CT) or magnetic resonance (MR) imaging. Treatment specifics, disease outcomes, and toxicity were collected from the multidisciplinary medical record. Toxicity was collected and coded according to the Common Terminology Criteria for Adverse Events ver. 4.03. Response after neoadjuvant therapy was assessed using the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines ver. 1.1 [17].
2. Statistical analysis
Patients were grouped according to treatment strategy: upfront surgery followed by adjuvant therapy or neoadjuvant therapy before less aggressive, organ preserving surgery. Neoadjuvant therapy and adjuvant therapy recommendations were external beam radiotherapy (XRT) and chemotherapy determined by the histology of the primary tumor and discretion of medical oncologist. All patients undergoing neoadjuvant therapy had planned re-imaging 4–8 weeks after XRT. Pathology was reviewed in all patients undergoing surgery. Tumor response to neoadjuvant therapy was scored as: <5% viable tumor, large amounts of necrosis with active tumor still present, or active tumor with no or minimal necrosis.
Loco-regional failure (LRF) was defined as pathologic or imaging findings consistent with tumor recurrence in the head and neck. Exenteration free survival (EFS) was defined as the time from diagnosis to orbital exenteration or death. Progression-free survival (PFS) was defined as the time from diagnosis to disease progression or death. Overall survival (OS) was defined as the time from diagnosis to death from any cause. Data analysis was performed using SPSS ver. 21 (IBM, Armonk, NY, USA). Kaplan-Meier methods were used to estimate survival. The log-rank test was used to compare groups. Logistic regression analysis and Cox proportional hazards modeling was used to determine predictors of LRF and survival. Chi-square testing was used to compare categorical variables. All analyses performed were two sided and considered significant at p ≤ 0.05.
Results
Twenty patients with orbit invasive sinonasal cancer determined to require an orbital exenteration at time of surgery were included. Fourteen patients underwent OP. Neoadjuvant treatment consisted of concurrent CRT in all patients aside from two who refused chemotherapy and underwent XRT alone. Surgery was performed a median of 60 days (range, 35 to 98 days) after completion of neoadjuvant CRT. Eight patients had squamous cell carcinoma, 4 patients had sinonasal-undifferentiated carcinoma, 2 patients had adenocarcinoma, 2 patients had mucoepidermoid carcinoma and 4 patients had other tumor types. The majority of patients received cisplatin chemotherapy concurrently with radiotherapy as part of their neoadjuvant treatment. Nine patients received weekly cisplatin and 2 patients received cisplatin at dose of 100 mg/m2, 1 dose at the beginning of therapy and again at the beginning of the third week.
Radiation therapy was given daily in 1.8–2.0 Gy fractions to 50 Gy or 50.4 Gy. A representative preoperative radiotherapy plan is shown in Fig. 1A. Three patients, all of whom received primary surgery including orbital exenteration followed by adjuvant CRT, received three-dimensional conformal radiotherapy (3D-CRT). Seventeen patients, including all patients treated with the OP approach received intensity-modulated radiation therapy (IMRT). Clinical target volume (CTV) encompassing subclinical disease was defined by a 7-mm margin added to either the gross target volume (GTV) consisting of the gross tumor or postoperative tumor bed. For patients treated with cervical lymph node involvement, CTV was extended to include the level II-IV neck bilaterally. An additional 5-mm margin was added to define the planning target volume (PTV), accounting for any possible deviations in plan targeting and delivery.
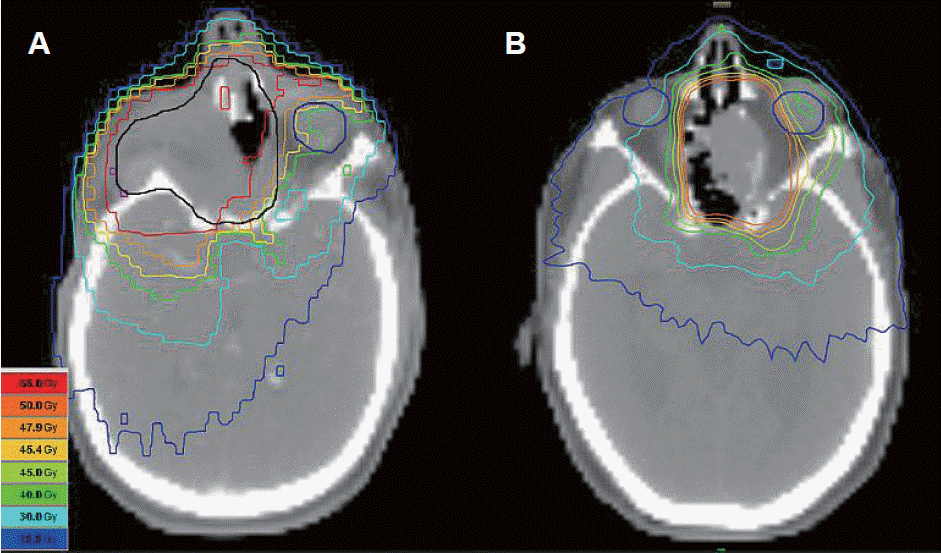
Representative postoperative intensity-modulated radiotherapy (IMRT) plan treated to 66 Gy (A) and preoperative IMRT plan treated to 50.4 Gy (B).
Six patients received primary surgery followed by adjuvant CRT. Most patients (66.7%) received carboplatin and paclitaxel chemotherapy following surgery. Radiation therapy was given daily in 1.8–2.0 Gy fractions to 66 Gy or 66.6 Gy. A representative postoperative radiotherapy plan is shown in Fig. 1B. Characteristics of patients in both groups are shown in Table 1.
Treatment response was evaluated 4 to 8 weeks following the completion of neoadjuvant therapy. Two patients did not undergo definitive surgical resection following neoadjuvant therapy. One patient developed renal failure during neoadjuvant CRT and expired and 1 patient refused surgical resection. Based on imaging, 3 patients were found to have a complete response, 5 patients were found to have a partial response, and 4 patients were found to have stable disease. Seven patients had either pathological no viable tumor remaining or less than 5% viable tumor at time of surgical resection. Another 2 patients had large amounts of necrosis, with some viable tumor remaining.
No patient required an orbital exenteration at time of definitive surgery following neoadjuvant therapy. All patients who did not receive neoadjuvant therapy underwent an orbital exenteration as part of upfront surgery. One patient who received neoadjuvant therapy required an orbital exenteration as salvage for a local-regional recurrence 35 months following surgery. Two-year EFS was 62.3% (95% confidence interval [CI], 36.7% to 100.0%) for patients receiving neoadjuvant therapy (Fig. 2).
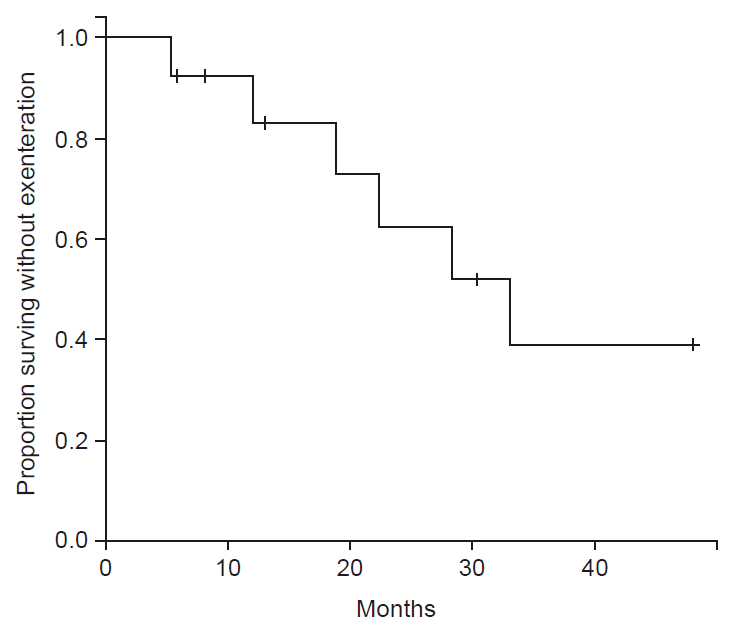
Kaplan-Meier estimate of exenteration free survival at intervals of time measured in months, in patients treated with neoadjuvant chemoradiotherapy (organ preservation approach only).
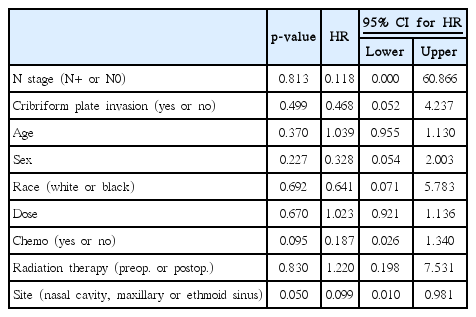
With a median 18.8 months of follow-up for patients, 2 of 6 patients receiving upfront surgery and 4 of 14 patients receiving OP with neoadjuvant therapy developed a local or regional recurrence. The 2-year Kaplan Meier estimate of locoregional control (LRC) was 75% for patients treated with initial surgical resection compared with 60% for patients treated neoadjuvant therapy (p = 0.997), shown in Fig. 3. Results of the Cox regression model are shown in Table 2. Primary site was correlated with local failure (p = 0.05). Freedom from LRF at 2 years was 85.7%, 80%, and 40% for maxillary sinus, nasal cavity, and ethmoid sinus tumor sites, respectively. Tumors originating from the nasal cavity had a lower rate of LRF (hazard ratio [HR], 0.099; 95% CI, 0.010 to 0.981) than ethmoid sinus and maxillary sinus tumors.
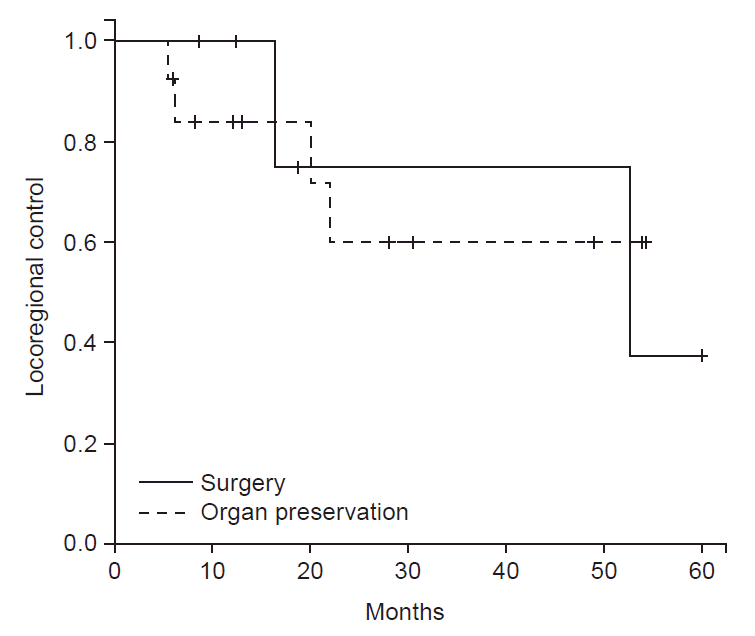
Kaplan-Meier estimate of locoregional control at intervals of time measured in months, stratified by treatment group (upfront surgery including orbital exenteration vs. organ preservation approach consisting of neoadjuvant chemoradiotherapy followed by less aggressive surgery not including orbital exenteration).
Thirteen patients had died at the time of analysis. Two year OS was 40% for patients treated with upfront surgical resection compared to 58% for patients who received OP and was not statistically different between treatment groups (p = 0.815). Two patients receiving OP died from distant metastases without LRC, and 1 patient receiving OP died from a second primary cancer (uterine). The lower rate of LRF observed in patients with the nasal cavity as their primary site may have translated into a trend towards reduced risk of death (HR, 0.240; 95% CI, 0.053 to 1.08).
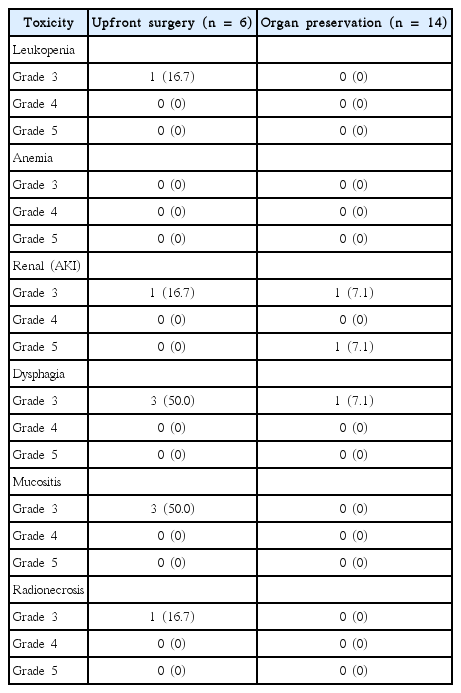
Treatments were generally well tolerated among patients in both groups. There was no surgical mortality, and no significant surgical toxicities were noted in the patients’ medical records. One patient receiving neoadjuvant CRT developed renal failure and died before completing the treatment course. No other grade 4 or 5 toxicities were seen. All patients in the orbital exenteration followed by adjuvant CRT treatment group and 4 patients undergoing OP experienced a grade 3 or higher toxicity. This difference was statistically significant between treatment groups (p = 0.003) favoring reduced toxicity with neoadjuvant treatment. Common toxicities for all patients are shown in Table 3.
Discussion and Conclusion
It has long been recognized that preservation of the orbital contents in locally advanced sinonasal cancer would be beneficial to patient’s quality of life and may be possible if surgery and postoperative radiotherapy were combined [10]. Our data demonstrates that preservation of the orbit with neoadjuvant CRT may be a successful treatment strategy in selected patients with orbit invasive sinonasal cancer. Furthermore, this approach may result in reduced toxicity for patients, while preserving oncologic outcomes.
For patients undergoing OP, EFS was high at 62.3% at 2 years. When considering all patients who were treated with an OP approach, only 1 (7.1%) required an orbital exenteration. This result compares favorably to other rates of OP in the literature, which range from 17%–100% [8,11,13,15]. Neoadjuvant therapy with CRT offers several distinct advantages over alternative OP approaches. It allows for a lower dose and smaller treatment volumes. Neoadjuvant radiotherapy also allows for assessment of treatment response at the time of definitive surgical resection.
In our study, despite complete radiologic response in only 21.4% of patients undergoing OP, half of patients had minimal viable tumor at time of surgical resection. This discrepancy demonstrates the difficulty of assessing disease response in the paranasal sinuses based on current imaging techniques and also suggests that doses of 50 Gy in standard fraction sizes are adequate preoperative therapy in these patients. The response rates appear similar to those previously reported. Kreppel et al. [18] published the results of 53 patients with locally advanced squamous cell carcinoma of the maxillary sinus and found a complete response rate of 21% after neoadjuvant CRT; higher response rates were seen with the use of cisplatin (10 of 11 patients with a complete response received cisplatin) [18]. However, despite the encouraging complete response rate, it is clear from other series of CRT alone that even with the administration of higher doses, a significant number of patients will ultimately fail loco-regionally [19-21]. Despite this, previous studies suggest patients who achieve a complete response after therapy appear to be in a better prognostic group than patients with a partial or stable response [5].
There were no significant differences between LRC, PFS, and OS between the two treatment groups but it does appear that despite treatment approach, outcomes for orbit invasive sinonasal cancers remain suboptimal. Although patients receiving OP had a numerically superior OS compared to those receiving initial orbital exenterations as part of surgical management despite having a numerically inferior rate of LRC, it is important to note these were non-statistically significant differences. In addition, due to the rarity of these tumors, our patient numbers were small. According to a recent meta-analysis by Reyes et al. [12] which considered 4 retrospective studies examining OP, no significant difference in survival was found when preserving the orbit [12]. In another large study examining rates of vision preservation, Sakashita et al. [13] did not find differences when preserving the orbital contents in terms of patient survival [13]. These previous studies agree with our findings that no significant differences in cancer specific outcomes or survival exist. In our study, patients with nasal cavity primaries were found to have significantly better LRC, which translated to a trend towards improved OS.
One barrier to OP has been the concerns of increased toxicity to either the eye’s visual function or from surgical morbidity from as subsequent surgery. Patients receiving preoperative therapy experienced less severe toxicity than those receiving postoperative therapy in our study. Three patients receiving postoperative therapy were treated with 3D-CRT instead of IMRT, which may have contributed to the increased rates of toxicity in this group. It is important to note patients receiving postoperative therapy in our study were treated to a higher dose of radiotherapy than those receiving preoperative therapy, and thus may have been at higher risk for toxicities including mucositis and dysphagia. This may be a potential advantage for using preoperative therapy to limit the risk for toxicity. As attempts have been made to preserve the orbit, higher rates of toxicity have been seen in patients undergoing irradiation after orbital surgery [8]. Neoadjuvant radiotherapy allows for smaller target volumes, significantly lower doses, and fewer complications from previous surgery. With the close proximity of sensitive organs at risk to target volumes in the sinonasal region, care must be taken when delivering therapy in the preoperative or postoperative setting. Increasingly conformal techniques such as intensity-modulated radiotherapy have helped to improve dose distributions in this region, resulting in a potentially improved toxicity profile [3,22].
Much of the published literature examining locally advanced sinonasal malignancy has limitations arising from the low annual incidence of this tumor class, heterogeneous tumor histology, and significant patient selection; our data is at risk for similar biases. This is a retrospective review of a small number of patients and care must be taken when applying its results to different patient populations. Additionally, differences may exist between treatment groups that our statistical methods did not have the power to detect given the relatively low patient numbers. Though no surgical mortalities were incurred, and no significant surgical toxicities were noted in the medical record, data regarding surgical toxicity was limited by the retrospective nature of study. Future studies will be needed in order to prospectively validate the success of an OP approach, however these studies may be difficult given the rarity of orbit invasive sinonasal tumors. Additionally lengthier follow-up will be needed to confirm long-term rates of OP and evaluate chronic toxicity rates of treatment.
In conclusion, locally advanced sinonasal cancer is an aggressive malignancy that often requires aggressive multimodality therapy, placing patients at risk for increased toxicity. Our data indicates that a strategy of OP with neoadjuvant CRT may be a successful approach that reduces toxicity while preserving outcomes in selected groups of patients with orbit invasive sinonasal cancer otherwise requiring an orbital exenteration. Additional studies will be needed to confirm these results, and evaluate long-term outcomes and toxicity in this patient population.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.