Evidence-based clinical recommendations for hypofractionated radiotherapy: exploring efficacy and safety - Part 1. Brain and head and neck
Article information
Abstract
Advances in radiotherapy (RT) techniques, including intensity-modulated RT and image-guided RT, have allowed hypofractionation, increasing the fraction size over the conventional dose of 1.8–2.0 Gy. Hypofractionation offers advantages such as shorter treatment times, improved compliance, and under specific conditions, particularly in tumors with a low α/β ratio, higher efficacy. It was initially explored for use in RT for prostate cancer and adjuvant RT for breast cancer, and its application has been extended to various other malignancies. Hypofractionated RT (HFRT) may also be effective in patients who are unable to undergo conventional treatment owing to poor performance status, comorbidities, or old age. The treatment of brain tumors with HFRT is relatively common because brain stereotactic radiosurgery has been performed for over two decades. However, re-irradiation of recurrent lesions and treatment of elderly or frail patients are areas under investigation. HFRT for head and neck cancer has not been widely used because of concerns regarding late toxicity. Thus, we aimed to provide a comprehensive summary of the current evidence for HFRT for brain tumors and head and neck cancer and to offer practical recommendations to clinicians faced with the challenge of choosing new treatment options.
Introduction
Radiotherapy (RT) delivers the total radiation dose divided into small daily fractional doses. Conventional fractionation typically involves a fraction size of 1.8–2 Gy, administered daily from Monday to Friday, five times a week. Various fractionation schemes have been investigated to improve the treatment outcomes. Hypofractionation was not widely adopted until the 1990s except for palliative purposes. Advances in technology have allowed the use of stereotactic body radiation therapy (SBRT) at selected sites such as the lungs, liver, and spine [1-5]. SBRT delivers a high ablative dose in fewer than five fractions, resulting in excellent local control. However, its use is limited to small-volume tumors and solitary or oligo-lesions, owing to the risk of toxicity.
Moderate hypofractionation was developed based on the results of randomized controlled trials (RCTs) for prostate and breast cancer [6-11]. Adjuvant hypofractionated whole-breast RT, consisting of 40 Gy in 15–16 fractions, demonstrated equivalent local control and fewer acute toxicities than the conventional RT schedule [6,7]. Moderately hypofractionated RT (HFRT) regimens for prostate cancer with a fraction size of 2.4–4 Gy over 4–6 weeks have shown non-inferior efficacy and similar toxicities in clinical trials [8-11].
Hypofractionation offers advantages, such as shorter overall treatment time, reduced cost, and improved patient compliance. For tumors with low α/β ratios, including breast cancer and prostate cancer, equivalent biological effects can be achieved at lower doses. Moderate hypofractionation is also advantageous for treating large volumes, including lymphatic chains. Various hypofractionation regimens have been investigated for the treatment of various malignancies. In addition to technological and theoretical advancements, the impact of coronavirus disease 2019 has accelerated the trend toward hypofractionation [12].
Thus, the demand for hypofractionation has increased. Advances in chemotherapeutic agents, targeted agents, and immunotherapy have led to longer life expectancies, resulting in more elderly patients, more cases requiring reirradiation, and the need for prolonged local control of metastatic disease. Despite the increasing attention to hypofractionation, determining the optimal scheme and indications remains a challenge. The lack of clinical trials and heterogeneity of published studies make it difficult to apply hypofractionation in daily practice.
To address these needs, we reviewed the current evidence on HFRT to clarify its efficacy and toxicities, organized by treatment site, in four parts. The first part of our review focuses on studies related to brain, head, and neck cancers. PubMed and ClinicalTrials.gov databases were searched for relevant articles written in English. Combinations of the following Medical Subject Headings terms including entry terms of each heading term were used: “radiation dose hypofractionation,” “glioblastoma,” “aged,” “frail elderly,” “meningioma,” “re-irradiation” for brain part, “radiation dose hypofractionation,” “head and neck neoplasms,” “aged,” “frail elderly,” “radiosurgery” for head and neck part. RCTs, meta-analyses, reviews, retrospective studies, and cohort studies, but not abstracts, were included. The literature search was performed on August 1, 2023. A consensus on the manuscript was reached by a group discussion of the subcommittee on hypofractionated radiation treatment guidelines of the Korean Society for Radiation Oncology.
Brain KQ1. What is the Optimal HFRT Schedule in Elderly Glioblastoma Patients?
Glioblastoma (GBM) is the most common malignant brain tumor. Its incidence is higher in patients aged >65 [13]. Elderly patients usually have additional comorbidities associated with a worse prognosis and less tolerance to toxicities [14]. The standard treatment for GBM typically involves a combination of surgery, RT, and chemotherapy [13,15]. In elderly patients, the choice of treatment strategy can be influenced by several factors, including overall health, functional status, tumor characteristics, and comorbidities.
For GBM, HFRT aims to complete the course of RT more quickly, which can be advantageous for elderly patients who may have limited tolerance for a prolonged treatment course. Several clinical trials have investigated HFRT schedules in elderly patients with GBM; however, the optimal regimen remains uncertain. Here, we review the results of phase 3 clinical studies on HFRT in elderly patients with GBM (Table 1).
1. Total 40 Gy in 15 fractions (2.67 Gy/fx) (randomized phase 3 trial)
Roa et al. [16] prospectively evaluated 100 GBM patients aged >60 years and compared a conventional fractionated RT (CFRT) regimen of 60 Gy over 6 weeks with an HFRT schedule of 40 Gy in 15 fractions over 3 weeks. This study was performed at four Canadian regional cancer centers between 1996 and 2001, before the Stupp regimen became standard [15], and therefore chemotherapy was not allowed before or during RT. The median Karnofsky performance scale (KPS) score was 70. There was no difference in overall survival (OS) between the CFRT and HFRT groups (median: 5.1 months vs. 5.6 months; p = 0.57). However, the proportion of patients requiring an increased dosage of steroids was doubled in the CFRT arm compared with the HFRT arm (49% vs. 23%; p = 0.02).
The HFRT schedule was then tested with the concurrent use of temozolomide (TMZ). Perry et al. [17] performed a randomized phase 3 trial comparing HFRT/TMZ followed by 12 cycles of TMZ to HFRT alone in elderly (aged ≥65 years) patients with an Eastern Cooperative Oncology Group performance status of 0–2. A total of 562 patients with a median age of 73 years were enrolled between 2007 and 2013. The OS was significantly improved from 7.6 months to 9.3 months with the addition of TMZ (p < 0.001). Median progression-free survival (PFS) was significantly improved in the HFRT/TMZ arm (3.9 months vs. 5.3 months, p < 0.001). However, a slightly higher rate of hematological adverse events was observed in the concurrent treatment group. The quality of life was similar between the two groups.
2. Total 34 Gy in 10 fractions (3.4 Gy/fx) (randomized phase 3 trial)
The Nordic Clinical Brain Tumor Study Group performed a 3-arm prospective randomized phase 3 trial comparing CFRT (60 Gy in 30 fractions), HFRT (34 Gy in 10 fractions), and six cycles of TMZ alone in patients with GBM older than 60 [18]. A total of 291 patients were randomized between 2000–2009. Overall, there was no significant difference in the median OS between the 60 Gy and 34 Gy group (p = 0.24). However, for patients older than 70 years, OS was better with TMZ alone and the HFRT group compared to the CFRT group—for TMZ vs. CFRT: hazard ration [HR] = 0.35 (95% confidence interval [CI] 0.21–0.56), p < 0.001; for HFRT vs. CFRT: HR = 0.59 (95% CI 0.37–0.93), p = 0.02. Patients treated with TMZ alone who had MGMT (O-6-methylguanine-DNA methyltransferase) promoter methylation had significantly longer survival than those without MGMT promoter methylation—9.7 months (95% CI 8.0–11.4) vs. 6.8 months (95% CI 5.9–7.7); HR = 0.56 (95% CI 0.34–0.93), p = 0.02. The researchers concluded that both TMZ alone and HFRT should be considered standard treatment options for elderly patients with GBM, especially those older than 70 years.
3. Total 25 Gy in 5 fractions (5 Gy/fx) (randomized phase 3 trial)
Roa et al. [19] further evaluated a shorter course schedule of 25 Gy in 5 fractions over 1 week and compared it to 40 Gy in 15 fractions over 3 weeks. Between 2010 and 2013, 98 patients aged >65 years or frail (age >50 and KPS 50–70) patients were enrolled. There was no difference in OS and PFS between the two arms with a median survival of 6.4 months and 7.9 months for patients treated with 40 Gy and 25 Gy, respectively (p = 0.988). Global health-related quality of life and steroid dose were also similar between the two arms.
4. Other HFRT study with higher doses (phase 2 trial)
The brain study group of the Italian Association of Radiation Oncology performed a multicenter phase 2 study [20]. In contrast to previous studies, they also included younger patients with good performance status, with the belief that HFRT could also be an option with a better quality of life and cost savings. However, only 24 patients were enrolled over 5 years (2009–2014). The median age of the patients was 61 years. The maximum permissible tumor size was 4 cm, as measured preoperatively. High dose of 67.5 Gy in 15 fractions was prescribed on postoperative cavity and enhancing tumor with additional 5-mm margin, and 52.5 Gy in 15 fractions with additional 10-mm margin. Concurrent and adjuvant TMZ administration was permitted. The median OS and PFS were 15.1 months and 8.6 months, respectively. Six patients required higher dose of anti-edema drugs, and one case of radiation necrosis (4.2%) was observed.
5. Recommendation
A combination regimen of CFRT (60 Gy in 30 fractions) and concurrent and adjuvant TMZ has shown a survival advantage over RT alone and has now become the standard treatment for patients [15]. However, this combined treatment may not represent the optimal approach in elderly patients [13,14]. A subsequent trend benefit analysis demonstrated a decreasing benefit with increasing age, with HR of 0.80 for the 65–71 years age group (p = 0.340) [21]. Therefore, it is important to note that treatment decisions should be made on an individual basis, considering various factors such as the patient's overall health, tumor characteristics, and patient preferences.
The optimal HFRT schedule for elderly patients with GBM should be determined through discussion between the patient and their healthcare team, taking into account the available evidence, treatment goals, and potential risks and benefits. To date, 40 Gy in 15 fractions is the most commonly used dose and can be administered concurrently with TMZ.
However, in terms of biologically equivalent dose, since GBM cells are usually estimated to have an α/β ratio of 8 Gy, 40 Gy in 15 fractions are lower dose compared to CFRT (75 Gy vs. 53.42 Gy). Additionally, several retrospective studies have suggested the possibility of increased progression with lower doses of HFRT [22,23].
Other dose schedules require more prospective data to confirm the safety, effectiveness, and possibility of the concurrent use of TMZ. We also considered that TMZ alone, without RT, could be an effective option, especially in patients with MGMT methylation. Recently, the Korean Radiation Oncology Group (KROG) recently approved a multi-institutional phase 3 prospective randomized trial unpublished; KROG 21-11, comparing 60 Gy in 30 fractions to 40 Gy in 15 fractions in GBM aged ≥70 years with a fair performance status of KPS ≥60 [22]. In younger patients with a good performance status, HFRT may be an option with higher doses than those administered to elderly patients. However, because of the lack of clinical evidence compared with CFRT, this should be applied only in clinical trials or in selected cases.
Brain KQ2. What is the Optimal Indication and Schedule of HFRT in Meningioma?
Meningiomas are the most common primary tumors of the central nervous system in adults. These tumors were classified according to the World Health Organization grading system. Grade 1 meningiomas are considered benign tumors (65%–80%), grade 2 are atypical and more aggressive (20%–35%), and grade 3 anaplastic meningiomas (3%) are malignant [24]. Despite recent promising progress in elucidating the genomic landscape and underlying biology of these histologically, molecularly, and clinically diverse tumors, the mainstay of meningioma treatment remains maximal safe resection while considering RT. When RT is required, it can be delivered as a single-fraction stereotactic radiosurgery (SRS), HFRT, or CFRT.
1. Grade 1 meningioma
Since the introduction of SRS around 1990, many studies have evaluated the use of single-session SRS for grade 1 meningiomas. The largest report on SRS for meningioma included 4,565 patients with 5,300 tumors treated at 15 European centers [25]. The median tumor volume was 4.8 mL, and the median dose was 14 Gy. The volume decreased in 58% of cases, with 5- and 10-year local control (LC) rates of 95.2% and 88.6%, respectively. Only 2.2% of progressive tumors require further treatment.
However, this effective SRS should be applied with caution in large and/or perioptic tumors because of the risk of edema, radiation necrosis, or neuropathy [26,27]. In such cases, HFRT can be adopted. Several studies have evaluated HFRT for meningiomas, which are usually limited to grade 1 meningiomas. Nguyen et al. [28] performed a systematic review and meta-analysis of HFRT for meningioma. They defined HFRT as delivering multifractionated photon RT with a prescription dose of ≥2.5 Gy per fraction, using either a linear accelerator-based platform, CyberKnife (CK) (Accuray Inc., Sunnyvale, CA, USA), or multisession Gamma Knife. The most commonly described doses were 25–30 Gy in 5 fractions and 15–21 Gy in 3 fractions. Overall, 14 studies involving 630 patients were analyzed. With median follow-up time from 24.5 to 57.5 months, the LC rate was 90%–100%, and median late toxicity rate was 10%. Of 474 tumors assessed for radiologic response, 78% were stable, 18% decreased in size, and 4% increased in size. Of 327 patients who were symptomatic prior to treatment, 17% had a complete resolution of symptoms, 41% showed improvement, 36% showed stability, and 6% showed deterioration after RT.
Recently, a prospective phase 2 study which evaluated the role of HFRT for large (>3 cm) or critical site (<3 mm from critical structures) meningiomas was published [29]. The study included 178 patients, and 25 Gy in 5 fractions was delivered at median 80% isodose line with CK (Accuray Inc.). After a median of 53 months, the 5-year LC rate was 97%, and neurological symptoms was improved in most of the patients. At the 5-year minimum follow-up, the toxicity rate was 11.7% (9 of 77 patients). The main toxicities were trigeminal numbness and visual impairment, with overall rates of 4% and 5%, respectively.
2. Grade 2, 3 meningioma
Compared with grade 1 meningiomas, clinical data on SRS or HFRT for grade 2 or 3 meningiomas are limited [24,30]. Usually, in grade 2 or 3 meningiomas, the target of RT includes not only the gross tumor, but also the surrounding normal tissue, which can harbor microscopic disease. A recently published contouring guideline suggests that the clinical target volume (CTV) should include an additional 5-mm and 1-cm margin to gross tumor volume (GTV) in grade 2 and 3 meningiomas, respectively, to include the preoperative tumor bed, peritumoral edema, hyperostotic bone changes, and dural enhancement or thickening as seen at diagnosis [31,32]. Therefore, grade 2 or 3 meningiomas that require adjuvant RT are typically treated with CFRT. Recently, the Radiation Therapy Oncology Group (RTOG) 0539 study results were published using high-dose intensity-modulated RT (IMRT) (54–60 Gy) with conventional margins (up to 2 cm) [33]. In this study, the 3-year PFS rate was 58.8%, and 92.9% of recurrences occurred within the planning target volume (PTV). However, there have also been some studies on the efficacy of HFRT for grade 2 and 3 meningiomas.
Marchetti et al. performed a phase 2 trial of the efficacy of HFRT for residual or recurrent grade 2 meningiomas [34]. The delivered dose was 24–28 Gy in 4 fractions with CK. Only enhanced tumors were treated. Twenty-four patients were enrolled, and after median follow-up of 28 months, eight patients (33.3%) had disease progression, resulting in a 3-year PFS of 47%. The in-field 3-year PFS was 86%.
Zhang et al. [35] also prospectively collected data from 44 patients (63 grade 2 lesions) and nine patients (16 grade 3 meningioma lesions) treated with SRS and HFRT. The lesions were treated in fractions for 1–5 sessions, with a median of one and three sessions for grade 2 and 3 lesions, respectively. The median dose was 20 Gy (range, 12 to 40 Gy). The 3- and 5-year PFS was 49% and 36% for grade 2 tumors, and 43% and 14% for grade 3 tumors, respectively.
Recently, Gagliardi et al. [30] performed a meta-analysis to evaluate the efficacy of RT and SRS/HFRT for grade 2 and 3 meningiomas. A total of 42 papers with 2,853 patients and 3,077 lesions were included in the analysis. The overall recurrence rate was 38% in the adjuvant CFRT group compared to 25% in the SRS/HFRT group (p = 0.01). However, the median time to recurrence was 1.5 times longer in the CFRT group, as well as the follow-up time, which makes the 5-year LC rate higher in the RT group (55% vs. 26%, p = 0.01).
3. Recommendation
Therefore, HFRT may be a useful treatment option for grade 1 meningiomas. Single-session SRS has been proven as effective as surgery for small, non-critical site meningiomas, with a great deal of evidence [24]. However, single-session SRS carries an increased risk of edema, brain necrosis, and neuropathic toxicities in certain cases [26,27]. Therefore, in these cases (large or critical site located), HFRT, usually with 25–30 Gy in 5 fractions or 18–21 Gy in 3 fractions, can be an effective option. Several retrospective and prospective trials have been published, although no phase 3 randomized trial have compared HFRT with CFRT [28].
For grade 2 and 3 meningiomas, evidence regarding the adoption of SRS or HFRT over CFRT remains scarce. However, several retrospective and prospective trials have evaluated the efficacy and safety of SRS and/or HFRT, particularly for relatively small tumors [36,37] (Table 2). The main purpose was to increase the biologically effective radiation dose and local control. However, the reported out-of-field recurrence rate is not negligible [30]. Therefore, owing to the scarcity of studies, the superiority of SRS or HFRT over CFRT for high-risk grade 2 or 3 meningiomas remains unclear.
Brain KQ3. Is HFRT Feasible for Brain Re-irradiation in Recurrent Glioma?
Even after a combination of surgery, RT, and chemotherapy, most patients with GBM progress within 10–15 months [38]. However, there is still no confirmed salvage option, and some patients received re-irradiation. In addition, in low-grade gliomas, RT is a common first-line treatment option, and many patients experience recurrence, even though the time to progression is longer than that in GBM. Therefore, a substantial number of patients with low-grade glioma require considering re-irradiation [38]. However, there is no standard fractionation schedule for re-irradiation in patients with gliomas. Re-irradiation with various techniques with brachytherapy, SRS, HFRT, and CFRT has been reviewed elsewhere [39,40]. Here, we review some prospective phase 1 and phase 2 trials which evaluated the feasibility of HFRT for re-irradiation in recurrent glioma [41-46] (Table 3).
1. Phase 1 trials
Moller et al. [41] evaluated the safety of HFRT re-irradiation in relapsed high-grade glioma. In this dose-escalation phase 1 trial, there were four groups: (1) 35 Gy in 10 fractions with a small PTV (<100 mL); (2) 35 Gy in 10 fractions and 7 Gy boost with a small PTV (<100 mL); (3) 29.5 Gy in 5 fractions with a small PTV (<100 mL); and (4) 35 Gy in 10 fractions with a large PTV (100–300 mL). Thirty-one patients were included in the study. The median PFS was 2.8 months, and the median OS was 7.0 months. Seven patients (23%) were progression-free 10 weeks after treatment and they were considered assessable for late toxicity. Of these seven patients, five were treated in Group 1 (3.5 Gy × 10) and two were treated in Group 2 (4.2 Gy × 10). None of the patients in Groups 3 (n = 5) or Group 4 (n = 5) were progression-free at 10 weeks.
Gutin et al. [42] evaluated another HFRT schedule of 30 Gy in 5 fractions using bevacizumab (BEV). Patients with recurrent glioma received BEV every 2 weeks for 28 days. HFRT was administered after the first BEV cycle. The PTV margin was 5 mm from GTV. Twenty-five patients with a median age of 56 years were enrolled in this study. For the GBM cohort, overall response rate was 50%, 6-month PFS was 65%, and median OS was 12.5 months. No radiation-induced necrosis was observed.
The same group performed a phase 1 dose escalation trial in 3 fractions [43]. In this study, the recurrent tumor volume was less than 40 mL. Three-fraction HFRT was administered every other day, starting at 9 Gy per fraction, after two BEV doses. Dose escalation was possible up to 33 Gy in 3 fractions, with no symptomatic necrosis in any of the 15 patients.
2. Phase 2 trial
Navarria et al. [44] performed a single-arm prospective phase 2 study to evaluate the efficacy of re-irradiation recurrent high-grade glioma patients. Ninety patients (72% GBM) with median interval time of 24 months received re-irradiation as follows: (1) <2 cm target, 25 Gy/1 fraction; (2) 2–3 cm target, 37.5 Gy/5 fractions; and (3) >3 cm or critical site located, 49.5 Gy/15 fractions. The CTV was a contrast-enhancing tumor on magnetic resonance imaging plus an area of [11C]-methionine positron emission tomography uptake. Sequential chemotherapy after re-irradiation was delivered in 36 patients (40%). The median OS and PFS were 17 months and 13 months, respectively. Grade 2–3 radiation necrosis occurred in nine patients (10%) at a median time of 6 months (range, 2 to 14 months).
Recently, the results of the NRG Oncology/RTOG 1205 study, a randomized phase 2 trial comparing concurrent BEV and re-irradiation versus BEV alone for recurrent GBM, were reported [45]. Patients with recurrence outside the original RT field with at least 6 months RT interval were included. The re-irradiation dose was 35 Gy in 10 fractions. A total of 170 patients were included in this study. After a median 12.8 months follow-up, the median OS was 9.7 months for the BEV only group and 10.1 months for the BEV+RT group were not significantly different (p = 0.46). Using the McDonald criteria, the 6-month PFS was 29.1% and 54.3% in favor of BEV+RT (p = 0.001). There was a 5% rate of acute grade 3 or higher treatment-related toxicities, and no delayed high-grade toxicities. This study concluded that re-irradiation of 35 Gy in 10 fractions with BEV can improve the 6-month PFS rate, although this does not translate into an OS benefit.
3. Recommendation
No standard care exists for patients with recurrent glioma. Re-irradiation is increasingly used for recurrent gliomas with the aid of technological advances that have improved the therapeutic ratio [40]. A meta-analysis of 50 studies which evaluate the efficacy of re-irradiation for recurrent GBM showed 6- and 12-month OS as 73% and 36%, respectively [39]. Although there was no dose-response relationship in this study, a HFRT regimen with ≤5 fractions seemed to provide superior 6-month PFS (47% vs. 26%, p = 0.005). However, the feasibility of brain re-irradiation, especially the adoption of HFRT, depends on several factors, including the patient's overall health, location and size of the target area, interval since the previous radiation treatment, and dose and volume of the previous radiation.
It is important to note that brain re-irradiation always carries an increased risk of cumulative radiation-related toxicities such as radiation necrosis. In the respect of relatively few reported cases, radiation necrosis is usually observed in recurrent tumors that generally receive cumulative total equivalent dose in 2 Gy fractions doses >120 Gy and volumes >40 mL [40]. This risk is not reported to be higher in patients receiving BEV. Careful patient selection, treatment planning, and monitoring are crucial when considering HFRT for brain re-irradiation.
The decision to pursue HFRT for brain re-irradiation should be made on an individual basis, taking into account the patient's specific clinical circumstances and the potential benefits and risks. The most common HFRT regimens adopted in clinical studies includes 30–35 Gy in 10 fractions or 25–35 Gy in 5–6 fractions. Usually, only a small of (3–5 mm) CTV/PTV margin is used. However, before adopting HFRT, a multidisciplinary team, including radiation oncologists, neurosurgeons, and other specialists, should be involved in the decision-making process to ensure the best possible patient outcomes.
Head and Neck KQ1. What is the Appropriate Hypofractionation Schedule for Elderly Frail Patients with Head and Neck Cancer?
Head and neck cancer is a locally advanced disease in 60% of newly diagnosed patients. Multimodal treatment is the standard treatment for these patients; however, it is accompanied by considerable acute and long-term toxicities. Definitively aimed radiotherapy or chemoradiotherapy (CCRT) may cause severe mucositis, dysphagia, xerostomia, and dermatitis, leading to a poor general condition and impaired function. Elderly frail patients may have difficulties completing definitive targeted treatments.
Achieving local control is crucial for preserving organ function and quality of life in patients with head and neck cancer. Elderly patients require a treatment approach that offers prolonged local control, in addition to the limited toxicity that they can tolerate. However, clear recommendations have not been made for the elderly or frail patients who are unfit for standard treatment.
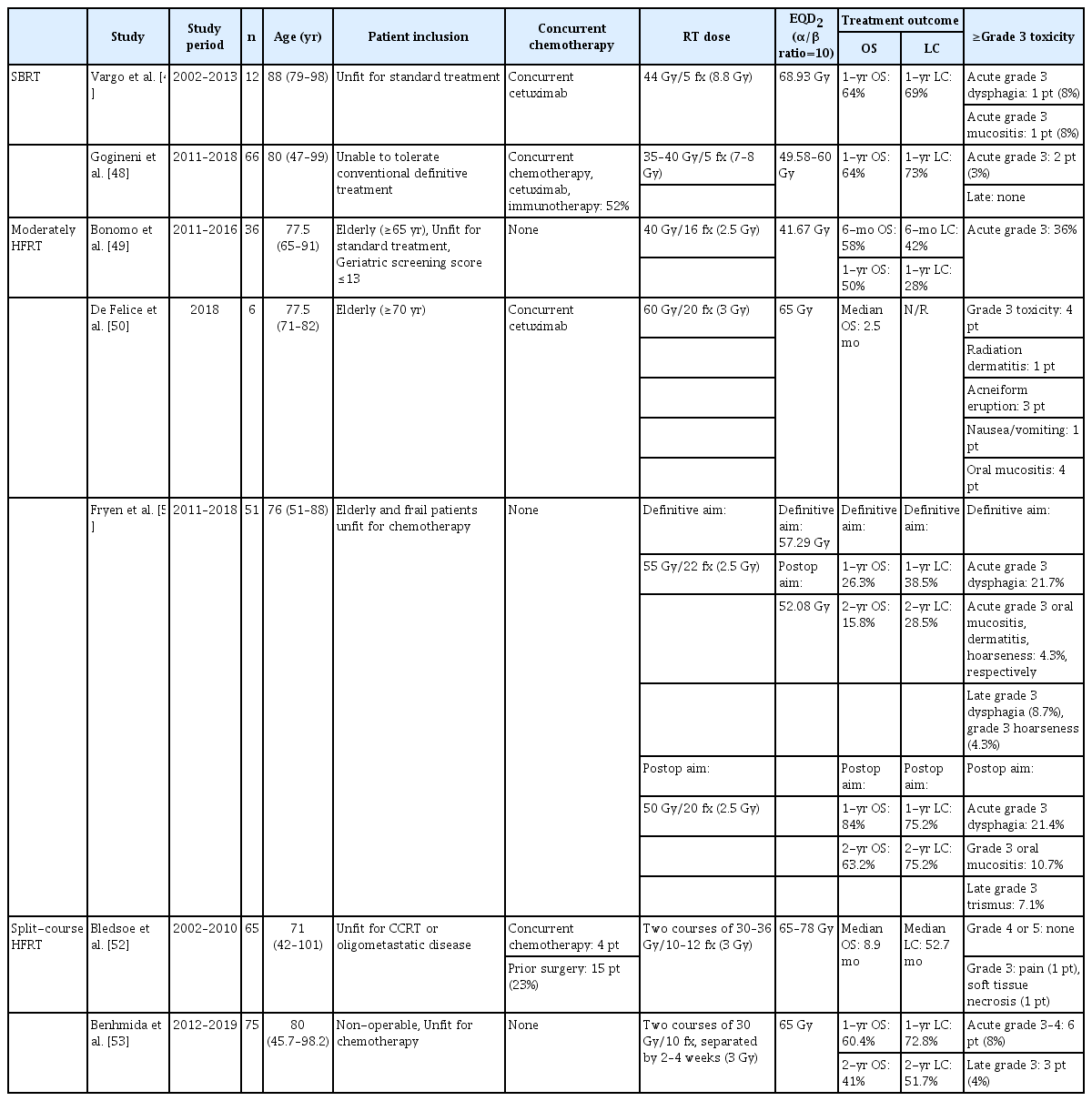
Although SBRT and HFRT have recently become more widely used, their application in head and neck cancers is limited owing to concerns regarding toxicity. The radiation field for head and neck cancer inevitably includes normal organs such as the salivary glands, oral cavity, esophagus, and mandible. SBRT or HFRT offers advantages such as shorter treatment time, less acute toxicity, and better compliance, but also has the potential to permanently damage organs. Several studies have reported on the efficacy and toxicity of SBRT and HFRT in elderly patients with head and neck cancer (Table 4).
1. SBRT
Vargo et al. [47] conducted a study involving 12 elderly patients with a median age of 88 years (range, 79 to 98 years) with medically inoperable head and neck cancer. These patients were selected based on the presence of a well-lateralized lesion and were unfit for standard treatment. The patients received SBRT with a total dose of 44 Gy delivered in 5 fractions on alternating days, along with concurrent cetuximab. The planned treatment was successfully administered to 92% of the patients. The study reported a 1-year LC rate of 69% and a 1-year OS rate of 64%. Acute grade 3 dysphagia and late grade 3 mucositis were reported in one patient each, with no occurrence of grade 4 or 5 toxicities.
In another study, 66 frail patients deemed unable to tolerate standard treatment for head and neck cancer were analyzed [48]. The median age of the patients was 80 years (range, 47 to 99 years). SBRT was delivered at a total dose of 35–40 Gy in 5 fractions to the GTV on a biweekly schedule. For patients with gross neck nodes, ipsilateral neck levels II–IV were included as the CTV, and a dose of 30 Gy in 5 fractions was delivered to the CTV. All patients completed SBRT. Concurrent immunotherapy, cetuximab, and chemotherapy were administered to 34 patients (52%). The study reported a 1-year LC rate of 73%, with a median time to local failure of 28.3 months. The 1-year OS rate was 64%. Two patients (3%) experienced grade 3 acute toxicity, including one patient with grade 3 dysphagia and one patient with grade 3 anorexia. No chronic grade ≥3 toxicities were reported.
2. Moderate HFRT
Moderately HFRT has been assessed in several studies using fraction sizes ranging from 2.5 to 3 Gy and a total of 15–20 fractions. Bonomo et al. [49] selected elderly patients who were unfit for definitive concurrent CCRT using the Geriatric 8 (G8) screening tool and Charlson Comorbidity Index. A G8 score of less than 14 was considered the cutoff value for frail elderly patients. The administered RT dose was 40 Gy in 16 fractions with a fraction size of 2.5 Gy. Out of a total of 36 patients, 33 patients (91.6%) completed the planned RT in 16 fractions. The objective response rate (ORR) was 66.6%, consisting of four complete responses (12.1%) and 18 partial responses (54.5%). The 1-year local-regional control (LRC) rate was 28% and the 1-year OS rate was 50%. Acute grade 3 mucositis was observed in seven patients (19.3%) and dysphagia in six patients (16.7%).
De Felice et al. [50] investigated a schedule of 60 Gy in 20 fractions for elderly patients aged 70 years or older. Six vulnerable patients who completed planned RT were included in this study. Three patients showed a partial response, and three patients had progressive disease 3 months after completing RT. The median PFS was 2 months and median OS was 2.5 months. More than grade 3 toxicities occurred in four patients, including radiation dermatitis in one, acneiform eruption in three, nausea/vomiting in one, and oral mucositis in four.
Fryen et al. [51] enrolled patients who underwent definitive and postoperative RT. A fraction size of 2.5 Gy was administered to all the patients. In the definitive RT group, the prescribed RT dose was 55 Gy for gross lesions and 45 Gy for both neck lymphatic chains. In the postoperative RT group, the prescribed RT dose was 50 Gy to the tumor bed and high-risk neck level and 45 Gy to the low-risk neck lymphatics. In the definitive RT group, the 1-year and 2-year OS rates were 26.3% and 15.8%, respectively. The 1-year and 2-year LRC rates were 38% and 28.5%, respectively. In the postoperative RT group, the 1-year and 2-year OS rates were 84% and 63.2%, respectively. The 1-year and 2-year LRC rates were both 75.2%. No grade 4 or 5 adverse events were reported. The incidence of grade 3 acute dysphagia was 21.7% in the definitive RT group and 21.4% in the postoperative RT group. In the definitive RT group, grade 3 late dysphagia was reported in two patients (8.7%) and grade 3 hoarseness in one patient (4.3%). In the postoperative RT group, grade 3 osteonecrosis occurred in two patients (7.1%).
3. Split-course HFRT
Bledsoe et al. [52] conducted a review of 65 patients who received split-course accelerated HFRT. Split-course RT was delivered in two courses of 30–36 Gy in 10–12 fractions. Concurrent chemotherapy was administered to four patients, and 15 patients (23%) underwent surgery prior to RT. Fifty-eight patients (89%) completed both courses of treatment. The median locoregional control was 25.7 months, and the median OS was 8.9 months. One patient experienced grade 3 or higher pain, and one patient had pharyngeal wall ulceration.
In another study by Benhmida et al. [53], patients were also treated with two courses of 30 Gy in 10 fractions separated by a mid-course break of 2–4 weeks. A total of 75 patients with a median age of 80 years were included in this study. The 1-year and 2-year LC rates were 72.8% and 51.7%, respectively. The 1-year and 2-year OS rates were 60.4% and 41%, respectively, with a median OS of 19.3 months. Acute grade 3 or higher toxicities occurred in six patients (8%), and late grade 3 or higher toxicities occurred in three patients (4%).
4. Recommendation
Although there is no well-designed prospective study, SBRT with 35–40 Gy in 5 fractions has shown acceptable efficacy and toxicity in elderly patients with well-lateralized gross tumors. Moderate hypofractionation (with 2.5–3 Gy fraction up to 55–60 Gy) and split-course hypofractionation (two courses of 30–36 Gy/10–12 fractions) treatments can be attempted in frail elderly patients unfit for high-dose definitive CCRT. The 1-year OS rate varies from 25% to 60% according to the tumor volume and location. However, all these studies were retrospective, and encouraging LC and OS have not yet been reported.
Head and Neck KQ2. What are the Appropriate SBRT Schedules and Indications for Re-irradiation in Cases of Recurrent Head and Neck Cancer?
Locoregional recurrence remains a significant challenge in the management of patients with head and neck cancers. Salvage surgery is the preferred treatment option for recurrent tumors in previously irradiated areas; however, it may not be suitable for patients with unresectable lesions or poor performance status. Systemic chemotherapy and immunotherapy have improved OS in such cases, but have not provided satisfactory locoregional control.
Recent advances in radiation techniques, particularly IMRT and SBRT, have garnered interest in reirradiation of recurrent head and neck cancer lesions. SBRT's ability of SBRT to deliver high-fractional radiation doses in a short treatment time offers potential benefits for overcoming tumor resistance and achieving locoregional control even at a relatively low total dose.
SBRT has shown great promise for treating various malignancies such as lung, spine, and liver tumors. Despite these encouraging outcomes, the use of SBRT for head and neck cancers has been approached with caution for several reasons. The head and neck region possesses a complex anatomy with numerous radiosensitive structures, and its response to high doses per fraction of SBRT remains uncertain. In the current standard of care, CFRT has been successful in achieving disease control in head and neck cancers, and increasing the dose per fraction may lead to a higher incidence of late toxicities. In addition, many head and neck cancers tend to spread to the neck lymph nodes and are conventionally treated electively. Incorporating elective nodal radiation into SBRT has the potential to increase late toxicity (Table 5).
1. Dose schemes investigated in studies
The first trial investigating the efficacy and safety of SBRT for recurrent head and neck cancer was conducted by Heron et al. [54] in 2009. The phase 1 dose-escalation study treated 25 patients in 5 fractions, delivering radiation doses from 25 Gy to 44 Gy over 2 weeks. No grade 3 or 4 adverse events or dose-limiting toxicities were observed. The ORR was 17.4%, and median duration of response, including the unconfirmed responses, was 3 months, with a maximum of 4 months. The 6-month disease-free rate was 31%, and the median OS was 6 months.
In a subsequent report by the same authors, the efficacy of cetuximab concurrent with SBRT was assessed in a retrospectively matched case-control study [55]. This study compared patients treated with SBRT alone with those treated with SBRT and concurrent cetuximab. The median SBRT dose was 40 Gy in 5 fractions. The results demonstrated improved 2-year locoregional control rates (33.6% vs. 49.2%, p = 0.009) and 2-year OS rates (21.1% vs. 53.3%, p = 0.031) in the SBRT with concurrent cetuximab group. No grade 4 or 5 adverse events were observed, and the incidence of grade 1–3 events did not differ significantly between the two groups. Only three patients experienced acute grade 3 toxicities (4.3%): one in the SBRT group had xerostomia, while two in the SBRT with concurrent cetuximab group had dysphagia and xerostomia, respectively.
Subsequent studies on SBRT for recurrent head and neck cancers were mostly retrospective in design [56-61]. The dose regimens varied, but most studies used doses ranging from to 35–45 Gy in to 4–5 fractions. In a multi-institutional study comparing 197 patients treated with SBRT and 217 patients treated with conventional IMRT [56], the median SBRT dose was 40 Gy in 5 fractions. The 2-year OS rate in the SBRT group was 16.3%. Subgroup analysis based on the recursive partitioning analysis (RPA) class was performed. RPA class I comprises patients >2 years from the first treatment with resected tumors; class II, those >2 years from the first treatment with unresected tumors or <2 years from the first treatment without organ dysfunction (defined as pretreatment feeding tube or tracheostomy use); and class III, those ≤2 years from the previous course with organ dysfunction. The subgroup analysis revealed that patients with RPA class III did not show any difference in OS between SBRT and conventional IMRT. The survival outcomes of patients with RPA class II disease are more complex. For small tumors such as volume less than 25 mL or rT0-2 tumors, SBRT with a dose of ≥35 Gy showed similar OS with IMRT with a median dose of 60 Gy, but SBRT with a dose of <35 Gy showed lower OS, though the difference was not statistically significant (50.3% for IMRT vs. 14.4% for SBRT <35 Gy vs. 38.5% for SBRT ≥35 Gy). However, for large tumors, such as volume more than 25 mL or rT3-4 tumors, SBRT exhibited inferior OS compared to IMRT, regardless of the total SBRT dose. Grade ≥3 acute and late toxicities were similar between the SBRT and IMRT groups.
Another recent phase 1 trial investigated the efficacy of SBRT with concurrent cisplatin [62]. The SBRT dose was elevated stepwise as follows: 30 Gy in 5 fractions, 35 Gy in 5 fractions, and 40 Gy in 5 fractions. Cisplatin was administered before every SBRT fraction at a dose of 15 mg/m2 for a total dose of 75 mg/m2 over the course of the SBRT treatment. After treating 18 patients, the 2-year OS rate and 2-year LC rate were 22% and 39%, respectively. Grade ≥3 acute toxicities were observed in 28% of patients. Three patients experienced grade 3 late toxicities, including dysphagia, aspiration pneumonia, and bleeding. No dose-limiting toxicities defined as grade ≥4 toxicities occurred in the 40 Gy in 5 fraction group. In the post hoc analysis, patients receiving 40 Gy had improved locoregional control compared to those receiving <40 Gy (locoregional failure: 33% vs. 100%, p = 0.023).
2. Factors to consider when deciding SBRT
Patient selection remains crucial for successful SBRT as a re-irradiation tool. SBRT may benefit patients with an anticipated poor prognosis, owing to its short treatment time. However, to achieve prolonged local control, sufficient dose delivery and target volume should be considered when selecting SBRT. Previous studies have shown that doses >35–40 Gy in 5 fractions are associated with better locoregional control and OS. A dose of 40 Gy in 5 fractions was biologically equivalent to 60 Gy of conventional IMRT, with an a/b ratio of 10. Factors such as tumor location, adjacent critical organs, and tumor size should be considered when prescribing doses. Conventional IMRT may be a better treatment option if the lesion is not suitable for delivering a sufficient dose of IMRT.
The target volume is another factor that should be considered. Small-volume tumors could be more successfully controlled at the same dose level, allowing for the prescription of a higher dose with relatively less toxicity. A study by the University of Pittsburgh showed that tumors ≤ 25 mL had less locoregional progression compared with GTV >25 mL, and this result has been reproduced in the results of a multi-institutional study previously described [63]. Another study that analyzed 137 patients showed that a target volume of >20 mL was associated with a significantly worse OS [58]. Patients with tumor volumes ≤20–25 mL could be considered as SBRT candidates rather than conventional IMRT. However, tumor volumes of 20–25 mL would be too small to be used as an inclusion criterion. Most studies did not use tumor volume as an inclusion criterion or included tumors <60 mL for a single lesion and <100 mL for all lesions [58].
The most significant late complications of carotid blowout syndrome (CBS) were observed in cases of re-irradiation, particularly when the maximum dose to the carotid artery exceeded 34 Gy. For patients receiving radiation treatment for the first time, the maximum dose can reach 47 Gy, according to findings from the International Stereotactic Body Radiotherapy Consortium survey [64]. The proper selection of patients based on the extent of disease around the carotid artery is crucial for mitigating risks. Moreover, SBRT has other potential toxicities, such as bone and soft tissue necrosis, dysphagia, trismus, fibrosis, and brain necrosis.
The current limitation of SBRT lies in the development of tumor radio-resistance in patients with recurrent head and neck squamous cell carcinoma who have previously undergone irradiation. Although dose escalation has proven effective in improving outcomes, the next logical step seems to be the utilization of a radiosensitizer. Adding three doses of cetuximab has shown promising results, providing a 2-year LC advantage (49.2% vs. 33.6%, p = 0.009) and an OS benefit (24.5 vs. 14.8 months, p = 0.03) compared with the SBRT alone approach. Notably, there was no significant increase in grade 3/4 toxicities observed [55]. Currently, this strategy is being actively investigated in phase 2 clinical trials. To validate these findings, a prospective randomized clinical trial was conducted.
3. Recommendation
Various studies have demonstrated that a dose range of 35–50 Gy in 3–8 fractions in small volume (≤25 mL) achieves comparable and favorable local control with acceptable acute and late toxicities in the context of SBRT. The 1-year LC varies between 71% and 87%, and the 1-year OS ranges 60% to 78%, respectively. With advances in SBRT technology, the incidence of grade 3–4 toxicities has been reported to be less than 10%–15%. Late complications such as CBS are a major concern, particularly in cases where high doses and large volumes are irradiated with SBRT. Tumor volume, location, and prescription dose should be carefully considered before deciding whether to administer SBRT.
To date no prospective comparative study has directly compared SBRT with other re-irradiation techniques such as IMRT to determine which radiation modality provides better LC rates with fewer acute toxicity effects. Promising clinical trials combining SBRT with immunotherapy (immune checkpoint inhibitors or cetuximab) are currently underway. This represents a potential avenue for future research on SBRT.
Conclusion
For brain tumors, HFRT in elderly or frail patients and grade 1 gliomas has relatively sufficient evidence, including phase 3 RCTs. In contrast, there is no high-level evidence for HFRT in grade 2 and 3 meningiomas or recurrent glioma. HFRT has not been widely used for head and neck cancer, and the evidence is mostly retrospective. Concerns regarding late toxicity in normal organs make it difficult to treat head and neck patients with hypofractionated schedules. Nevertheless, the results of phase 1–2 trials and retrospective data are accumulating. Prospective multicenter studies should be conducted in the future.
Notes
Statement of Ethics
Because this study did not involve any human subjects, Institutional Review Board approval and informed consent were not required.
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Funding
None.
Author Contributions
Conceptualization: Kim YS, Sung SY; Investigation and methodology: Kim YS, Sung SY; Project administration: Kim YS; Resources & Writing of the original draft: Sung SY, Song JH; Writing of the review and editing: Kim YS, Kim BH, Kwak YK, Kim KS, Yoo GS, Byun HK, Kim YJ; Data curation: Kim BH, Kwak YK, Kim KS, Yoo GS, Byun HK, Kim YJ.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.