Clinical management of uveal melanoma: a comprehensive review with a treatment algorithm
Article information
Abstract
Uveal melanoma (UM), the most frequently occurring non-cutaneous melanoma and most common primary intraocular malignancy in adults, arises from the melanocytes of the choroid in approximately 95% of cases. Prompt diagnosis and treatment is vital as primary tumor size is one of the key factors associated with survival. Despite recent advances in management, more than half of the patients develop metastatic disease which portends poor survival. Currently, treatment options for UM include local resection, enucleation, plaque brachytherapy, and/or particle beam radiotherapy (RT). Enucleation was initially the standard of care in the management of UM, but a shift towards eye-preserving therapeutic choices such as RT and local resection has been noted in recent decades. Plaque brachytherapy, a form of localized RT, is the most popular option and is now the preferred treatment modality for a majority of UM cases. In this review we discuss the etiopathogenesis, clinical presentation and diagnosis of UM and place a special emphasis on its therapeutic options. Furthermore, we review the current literature on UM management and propose a functional treatment algorithm for non-metastatic disease.
Introduction
Melanoma is a malignancy that originates from the neoplastic proliferation of melanin-producing cells known as melanocytes, which can be primarily found in the skin, ocular region and mucous membranes. Uveal melanoma (UM) is the most frequently occurring non-cutaneous melanoma and is the most common primary intraocular malignancy in adults [1]. The uveal tract, a layer underlying the sclera of the eye, includes the iris, ciliary body and choroid. Around 95% of uveal melanomas arise from the choroidal melanocytes.
1. Epidemiology
The worldwide incidence of UM is estimated to be close to 4 to 5 cases per million per year and varies with gender, race and geographical location. Even though most studies reveal no apparent gender preponderance, some European studies have seen a slightly increased incidence in males [2]. Caucasians are most commonly affected ethic group, accounting for 98% of UM cases. An increase in incidence of UM with latitude has also been observed with a decreasing gradient of cases from Northern to Southern Europe being reported. Furthermore, lower incidences of UM have also been noted in Asian and African nations [3].
2. Etiopathogenesis
As with many other types of malignancies, the precise causative elements for this rare cancer have yet to be clearly established. Both genetic and environmental risk factors have been implicated in the etiology of developing UM. Genetic factors previously associated include fair skin, inability to tan and light eye color [4]. A positive family history of UM, increased frequency of oculodermal melanocytosis, BAP1 mutations and dysplastic nevi have also been linked to a higher incidence of UM [5].
Although sunlight exposure is an independent risk factor in the development of cutaneous melanoma, epidemiological attempts to analyze the association between exposure to ultraviolet (UV) light and UM have led to contradicting results. Some studies suggest that UV light exposure is a synergistic risk factor for developing UM in individuals with light iris color [6]. Mutation mechanisms with GNAQ and GNA11 signatures noted in illuminated areas of the uvea have also suggested that sunlight exposure may be an independent risk [7]. However, as previously stated, other attempts to associate intermittent and chronic UV light exposure with development of UM have led to inconsistent results [8]. The ability of the cornea and lens to filter a major portion of UV radiation in sunlight before it reaches the uveal tissue has been suggested as a possible explanation for this unclear association [9]. Finally, occupational hazards, such as welding, have also been associated as an etiologic source for developing UM as it usually involves additional chemical exposure along with both infrared and visible radiation.
3. Clinical features
Nearly half of the patients at the time of diagnosis are asymptomatic and UM is only identified after routine eye examination. Symptomatic patients present with ophthalmological features such as floaters, photopsia, visual field defects, metamorphopsia, visible tumor, and/or painless loss of vision [10]. Iris melanomas are diagnosed almost 15–20 years earlier than choroidal or ciliary body melanomas, primarily owing to early iris color changes and distortion of the pupil.
4. Diagnosis
There are a significant differential diagnoses of UM that include choroidal nevus, retinal pigment epithelium hypertrophy and disciform degeneration, making it is integral to obtain an accurate diagnosis as soon as possible. A fundoscopic examination is the first step in obtaining this goal. Presence of orange pigment, subretinal fluid, and documentation of tumor growth all aid in obtaining an appropriate diagnosis [11]. Further evaluation is needed for additional tumor characterization with procedures such as optical coherence tomography ultrasound and/or fluorescein angiography. Ultrasound features of UM include an intrinsic acoustic quiet zone and decreased internal reflectivity of the tumor. Visually, the tumor is often described as circumscribed mushroom or dome-shaped mass after rupture of the Bruch’s membrane of the retina [12]. Fluorescein angiography helps to evaluate the vascular supply of the tumor. A biopsy is not required for diagnosis, but is often performed for prognostic purposes. An understanding of the molecular structure of the tumor helps in risk stratification and determining initial treatment paradigms.
5. Prognosis
Despite advances in UM therapies, almost half of the patients are ultimately at risk for developing metastatic disease, primarily due to the inability to identify a unique feature of UM which is early micro-metastases [13]. Owing to this frequent subsequent presentation, patients diagnosed with UM require periodic surveillance with physical examination, blood tests and radiographic imaging such as CT, MRI, abdominal ultrasound or PET/CT. The most common initial distant metastatic sites include the liver followed by the lung, skin and bone. Ciliary body involvement, older age, epithelioid subtype and large tumor basal diameters are tumor features associated with poor prognosis [14]. Recent knowledge of detailed molecular mechanisms underlying UM has led to more accurate prognostic predictions. The 8q gain mutation has been associated with increased risk of metastasis while monosomy 3 has been associated with a decreased risk [15]. Additionally, gene expression profiling has proven to be a superior predictor of prognostic and metastatic potential in UM. Finally, detection of circulating tumor DNA is another important predictive factor for developing metastasis [16].
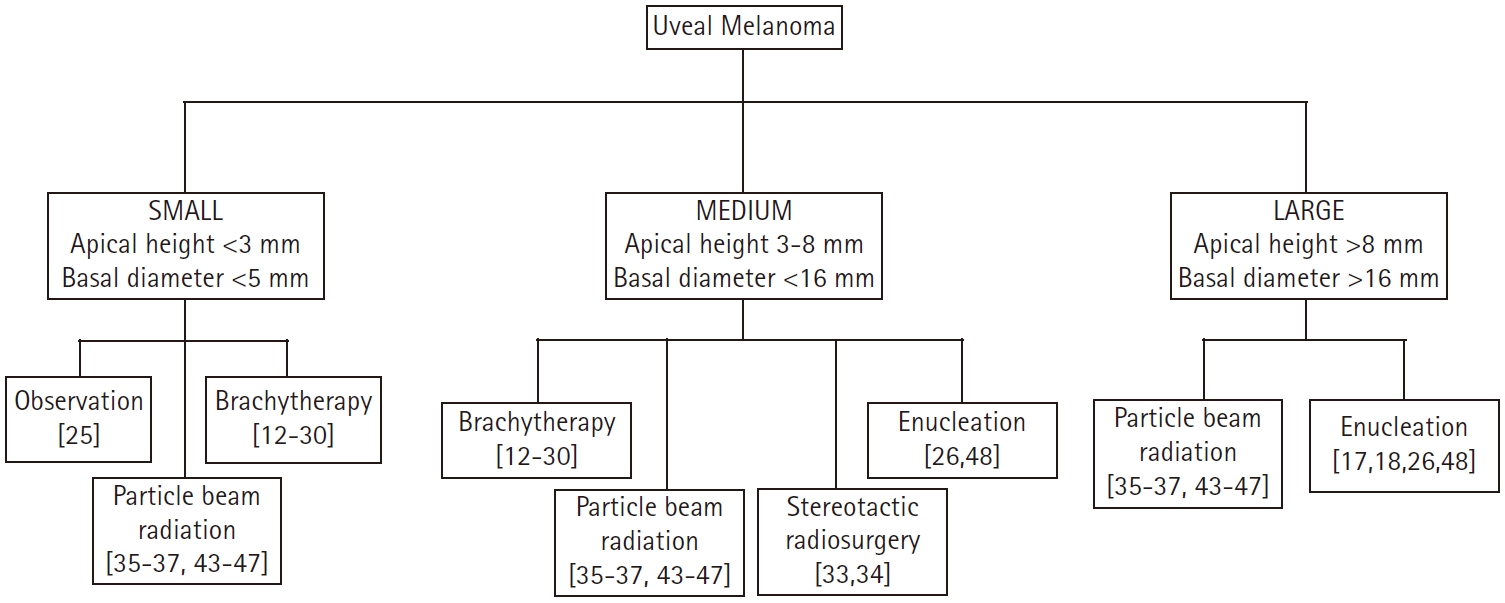
Given this constantly evolving treatment paradigm, herein, we evaluate the published data on local therapeutic options for non-metastatic UM and propose a functional treatment algorithm (Fig. 1).
Treatment Modalities
Most common treatment options for non-metastatic UM include surgery, plaque brachytherapy, and/or particle beam radiotherapy (RT). Surgical options include local resection, enucleation and orbital exenteration. Local resection can be achieved either by exoresection, which involves en bloc tumor removal via a scleral approach, or by endoresection, which is piecemeal removal via a vitreoretinal approach. These eye-conserving treatment approaches, in recent years, are now preferred over enucleation [17]. Enucleation is the surgical removal of the eye itself sparing the extraocular muscles and remaining orbital contents. Orbital exenteration involves the surgical removal of the entire orbital tissue including the eye, periorbita, appendages and eyelids. It is the preferred surgical approach for patients with large extraocular invasion or orbital extension. One added benefit of a surgical therapeutic approach is the ability to obtain adequate tissue samples for a detailed histopathologic and genetic analyses.
Plaque brachytherapy or plaque radiotherapy is the most widely used treatment modality in the management of UM and involves administration of a fixed dose of RT to the tumor. This is achieved by insertion of a radioactive implant into the episcleral tissue that delivers an apex RT dose of 80–100 Gy [18,19]. The most frequently employed radioisotope in the treatment of UM is Iodine-125 (125I) owing to its favorable dosimetric profile, followed by Ruthenium-106 (106Ru), and Palladium-103 (103Pd) [20]. The most common complications of brachytherapy administration are retinopathy, cataract formations, macular edema, neovascular glaucoma, dry eye, keratitis, eye pain, and scleral necrosis. Finally, improved early outcomes have been noted in UM patients when brachytherapy administration has been aided with the use of additional techniques such as intraoperative ultrasound guidance and echographic confirmation of plaque placement [21].
Particle beam therapy (PBT) or charged-particle radiotherapy (CPRT) is the second most frequently used form of RT in the treatment of UM. Protons, helium ions and carbon ions are delivered as highly precise external RT beams with a pre-specified dose. When PBT is utilized, a RT dose of 50–70 cobalt gray equivalent (CGyE) is usually delivered in 4 to 5 fractions. When carbon ions are used, a dose of 60–85 CGyE is delivered in 4 to 5 fractions. Owing to their physical properties, charged particles provide increased targeting, especially at the end of the beam range [22]. Furthermore, the usage of tantalum chips and volumetric planning in three dimensions also lead to optimal dosage administration. However, in spite of a precise homogenous dose delivery to the tumor, CPRT can also cause damage to the surrounding normal ocular structures leading to toxicities such as maculopathy, retinal detachment, glaucoma, cataract, vitreous hemorrhage and papillopathy [23].
Current Treatment Strategies based on Size of UM Tumor
The overall tumor size for UM is assessed based on both the apical height as well as the largest basal diameter of the tumor and is classified based on guidelines from the Collaborative Ocular Melanoma Study (COMS) group [24].
1. Small tumors
A landmark COMS study elucidated that tumors measuring <3 mm in apical height along with having a basal diameter measuring <5 mm should be primarily managed with observation [24]. There was no difference seen between patients enrolled in the study who received immediate therapeutic intervention versus those who pursued close observation and therefore the conclusion was to reserve treatment only at the time of tumor growth. Only 21% of patients on this trial demonstrated tumor growth in 2 years while 31% had tumor growth at 5 years post diagnosis.
2. Medium tumors
Medium tumors are defined as an apical height of 3–8 mm and a basal diameter of <16 mm. Treatment options for these patients range from plaque brachytherapy to PBT to enucleation. Another landmark COMS study, conducted over a span of 10 years, evaluated the quality of life after 125I plaque brachytherapy (IBT) or enucleation in 209 patients with choroidal melanoma. They concluded that there was no significant difference in survival between the two groups, but revealed that there was better visual function, defined as peripheral vision, for up to 2 years after treatment in patients who underwent IBT when compared to enucleation [25].
Several other studies have been conducted with an aim to explore the use of different radioisotopes in plaque brachytherapy for UM [26-30] (Table 1). Verschueren et al. [30], analyzed the long-term outcomes of 106Ru brachytherapy in 425 patients with small or intermediate UM. They observed a 5-year local control (LC) and overall control of 96% and 79.6%, respectively and also revealed functional and cosmetic eye preservation rates at 5 years of 52% and 96%, respectively. Takiar et al. [28] also demonstrated excellent tumor control and acceptable toxicity levels after 106Ru brachytherapy in a cohort study of 40 patients with UM. Actuarial 5-year LC and overall survival were 97% and 92%, respectively. Enucleation was not required in any of the patients and there was no diagnosis of neovascular glaucoma at follow-up. Tarmann et al. [29] evaluated the medical records of 143 patients managed with 106Ru brachytherapy for UM and demonstrated excellent rates for tumor control with a 2- and 4-year recurrence rate of 8.4% and 14.7%, respectively. They also revealed promising eye-preservation results with the likelihood of keeping the eye in 94.7% of the patients at 24 months and 91.8% at 48 months post-brachytherapy. 103Pd plaque brachytherapy was evaluated by Finger et al. [26] in a retrospective case series of 400 patients with UM. They concluded that 103Pd provided a superior option compared to alternative forms of radiation and demonstrated a local control of 96.7%; only 14 patients in the study required enucleation at a later date. Larger trials are needed to ascertain the optimal dosage of IBT in the treatment of UM.
Finally, more recent data is emerging looking at additional treatment options in conjunction with IBT. Use of intravitreal bevacizumab at the time of plaque removal and at 4-month intervals for a period of 2 years in 292 patients with UM showed significantly decreased macular edema and vision loss in these patients [31]. Additionally a prospective non-comparative interventional case series by Shields et al. [27] in 270 patients with choroidal melanoma studied the effectiveness of IBT followed by transpupillary thermotherapy (TTT). TTT is a non-invasive treatment option where infrared lasers are delivered to the tumor and is mostly effective for smaller low-risk tumors [32]. Shields et al. [27] demonstrated that plaque brachytherapy followed by 3 sessions of TTT resulted in a tumor recurrence of only 2% at 2-year follow-up and 3% at 5-year follow-up.
Stereotactic photon beam radiosurgery (SRS) is another option that can be employed in the treatment of UM in the medium-sized tumor group. Gamma knife, CyberKnife or linear accelerator platforms are some of modalities utilized [33]. Sikuade et al. [34] conducted a review of 191 patients with UM who were managed with either SRS (n = 85) or PBT (n = 106). They concluded that both treatments had excellent LC rates and eye preservation rates (98% and 95% of SRS and PBT groups, respectively), but there was superior visual prognosis in the PBT group when compared to SRS (65% vs. 45%; p = 0.008).
3. Large tumors
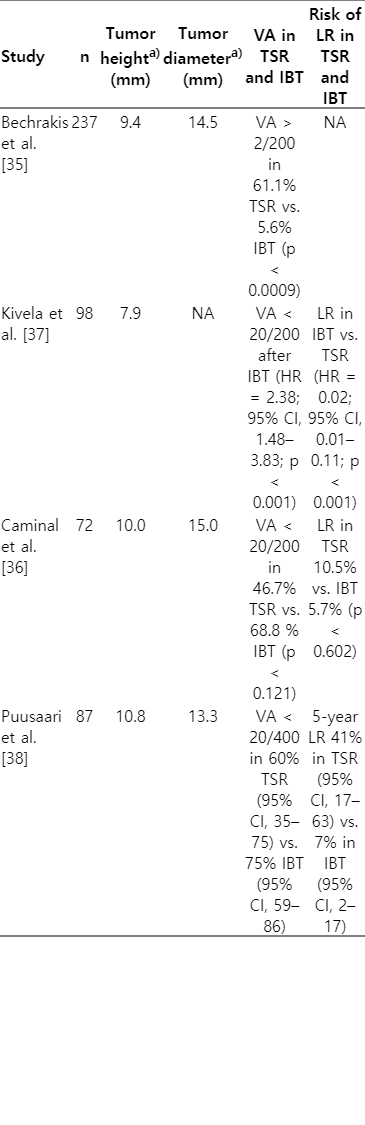
Several studies have been performed to compare the effectiveness of surgical procedures to brachytherapy in the management of large UM, defined as an apical height >8 mm or a basal diameter of >16 mm [35-38] (Table 2). The most notable therapies utilized are CPRT and enucleation. That being said, IBT is also considered a potential option for large tumors. A large retrospective, comparative, non-randomized study of 237 patients with large UM (defined as thickness >7.5 mm) by Bechrakis et al. [35], compared IBT to transscleral tumor resection (TSR) and demonstrated better visual acuity was retained in the TSR group (61.1% vs. 5.6%; p < 0.0009) as well as lower incidence of secondary glaucoma in TSR group when compared to IBT (5.6% vs. 33.3%; p = 0.03). There was no difference, however, in the mortality rates between the two groups. A matched case-control study by Kivela et al. [37], compared the complication rates, tumor control and visual acuity following IBT to TSR in 49 pairs of patients with large choroidal melanomas (thickness >6 mm). The results of this study suggested that the risk of losing 20/200 vision was higher after IBT compared to TSR (hazard ratio [HR] = 2.38; 95% confidence interval [CI], 1.46–3.83; p < 0.001) but there was a lower risk of tumor recurrence after IBT compared to TSR (HR = 0.02; 95% CI, 0.01–0.11; p < 0.001). A retrospective study by Puusaari et al. [38], in 87 patients with large UM compared TSR to IBT revealed promising results for improving visual acuity in the TSR group but also noted an increase in rates of local recurrence. They observed that the 2-year incidence of losing 20/400 vision was 60% (95% CI, 35–75) for TSR group and 75% (95% CI, 59–86) for IBT group but the risk of 5-year local recurrence in TSR group and IBT group was 41% (95% CI, 17–63) and 7% (95% CI, 2–17), respectively. The Zimmerman-McLean-Foster hypothesis suggests that rates of tumor recurrence is higher after surgical intervention of UM due to tumor manipulation during the procedures which may accelerate tumor cell dissemination [39,40]. Despite this hypothesis, given improved visual acuity and equivalent survivals, TSR has been advocated as an alternative to enucleation and RT in the treatment of large uveal melanomas [41,42].
Regarding CPRT, there are many studies detailing the use of PBT in comparison with other treatment modalities for UM [34,43-46] (Table 3). Abrams et al. [47] conducted a survival analysis of 1,004 cases of UM in which 380 cases were managed with external beam radiotherapy (EBRT) and 624 cases were managed with plaque brachytherapy. No difference in 5-year overall survival was seen between the two groups (83.3% EBRT vs. 82.5% BT; p = 0.69). Caujolle et al. [44] performed a retrospective study in 886 patients with UM who were managed with PBT They observed LC rates of 93.9% and 92.1% at 5 and 10 years, respectively and also noted ocular conservation rates of 91.1% and 87.3% at 5 and 10 years, respectively. A prospective, interventional, noncomparative study performed in 2645 patients by Egger et al. [45], analyzed eye preservations rates in patients managed with PBT and found that overall eye retention rates were 88.9%, 86.2%, and 83.7% at 5, 10, and 15 years, respectively. They concluded that favorable results were noted even for larger tumors and tumors near the optic disc. A retrospective, consecutive cohort study in 492 patients with large UM by Bensoussan et al. [43], noted good LC with overall and specific survival rates at 5 years of 65% and 75%, respectively. They concluded that PBT can serve as an effective alternative to enucleation in patients with large tumors. The UCSF-LBNL randomized trial by Mishra et al. [46] conducted in 184 patients with UM, included 86 patients receiving Helium ion particle therapy and 98 patients receiving IBT. They noted significantly improved LC in particle arm compared to the IBT arm (100% vs. 84% at 5 years, 98% vs. 79% at 12 years; log-rank p = 0.0006). Significantly lower need for further enucleation was also demonstrated in particle arm (11% vs. 22% at 5 years, 17% vs. 37% at 12 years; log-rank p = 0.01). Given numerous prior studies revealed promising results, CPRT is now used as a definitive treatment option in large tumors. Furthermore, CPRT gains an obvious advantage when the UM is in a circumpapillary location surrounding the optic nerve, as it is not feasible to place a plaque completely around the tumor and hence CPRT is preferred. Additionally, in large tumors where IBT is not appropriate, CPRT is the preferred treatment modality over enucleation when eye preservation is desired [48].
Finally, Bechrakis and Foerster [49] developed a novel approach of combining neoadjuvant PBT and subsequent endoresection in 58 patients with large UM (thickness >7 mm). They concluded that high-risk patients did not have increased morbidity and showed a lower rate of ocular side effects such as cataracts and retinal detachment in short-term follow-up compared to patients with UM of smaller size (thickness <7 mm). A retrospective interventional case series by Willerding et al. [50], also studied the benefits of neoadjuvant PBT prior to TSR in 106 patients with UM. Local recurrence was noted in 5 patients while enucleation was required in 10 patients. The study concluded that there were no significant risk factors noted for local recurrence but also stated that additional vitreoretinal surgery was frequently needed (69.8%).
In summary, surgical intervention such as enucleation is the preferred approach in large tumors which cannot be effectively managed with RT, especially if they are well-circumscribed or juxtapapillary in location. The use of neoadjuvant RT prior to performing surgery has been shown to produce improved results and a decrease in the potential risk of tumor seeding. Further evaluation of this combined modality approach is required but may be appropriate patients presenting with neovascular glaucoma, tumor replacing more than half of the globe, orbital invasion or optic nerve involvement.
Conclusion
UM, the most common primary intraocular malignancy, continues to provide daunting challenges in its treatment management. With more than half the patients developing metastatic disease after initial non-metastatic presentation, prompt diagnosis and treatment play a crucial role in alleviating the morbidity and mortality of this disease. Currently, RT is the most common treatment approach in the management of UM, especially for small and intermediate-sized tumors. IBT is the most frequently employed type of RT, followed by CPRT. The most common surgical approaches remain enucleation and local resection. In recent years, enucleation is considered as an option only in patients with large tumors or in those with optic nerve involvement where RT does not result in a favorable outcome. The popularity of local resection has also diminished of late, as RT provides a superior alternative; however, when local resection is performed as a primary treatment modality, it is often coupled with neoadjuvant or adjuvant RT to further decrease chances of local recurrence. Additional clinical trials and targeted therapies aimed at the molecular pathogenesis of UM may offer novel avenues in managing this disease in the future.
Notes
Conflict of Interest
No potential conflict of interest relevant to this article was reported.