Discussion
With certain exceptions, surgical resection with complete margins is ideal for most recurrent tumors with prior irradiation. Unfortunately, management can be challenging as surgical resection may not always be feasible due to potential morbidity. Radiotherapy is often considered for definitive treatment as an alternative, but the cumulative dose and subsequent late toxicities often prohibit the delivery of curative doses. Re-irradiation with PLDR is an attractive option in this setting. Although the mechanisms are incompletely understood, pre-clinical studies have demonstrated low-dose hyper-radiosensitivity of tumor cells, a phenomenon also known as the “inverse dose rate effect” [
6]. This phenomenon provides the potential for potentiating anti-tumor effects while minimizing normal tissue toxicity. The rationale behind this method lies in exploiting differences in radiation response between tumor cells and normal cells. As the radiation doses are delivered in pulses, allowing healthy cells to repair and recover between treatments. On the other hand, tumor cells are selectively damaged due to their reduced ability to repair DNA damage, thereby enhancing the therapeutic ratio in re-irradiation. There is also murine evidence that PLDR has better synergism with anti-programmed death-1 antibody than conventional radiotherapy [
7].
Clinical studies on PLDR are limited mainly to single-institution retrospective studies and are mostly concerned with intra-cranial tumors, which likely reflects the limited treatment options in locally recurrent brain malignancies [
8].
In a more recent report on novel unconventional radiotherapy techniques by Tubin et al. [
9], PLDR was noted as a novel technique to overcome radio-resistance and mitigate normal organ toxicities in re-irradiation. However, the authors noted that although retrospective studies and even a single-arm prospective trial have been published, comparative studies with conventional delivery are lacking [
9]. Additionally, many reports have limited follow-up. Thus, questions remain regarding the long-term effects of PLDR.
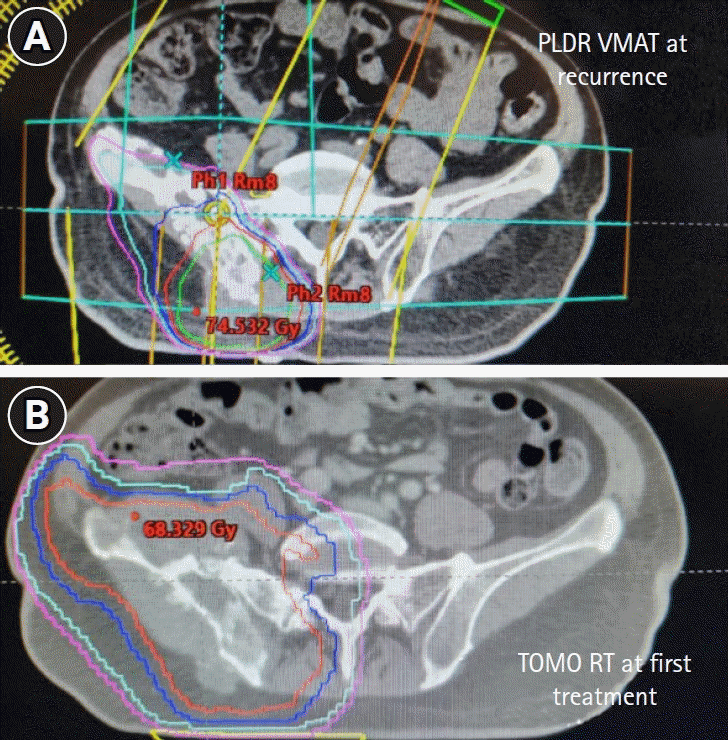
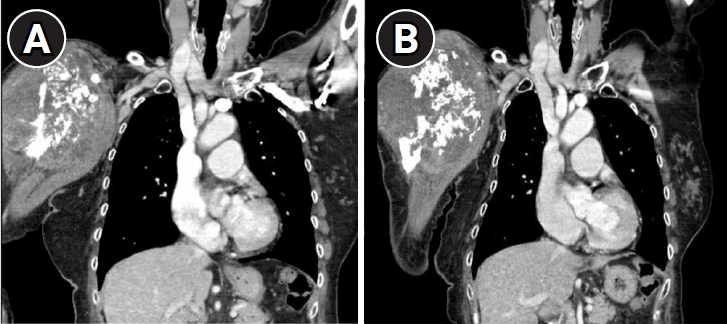
In this study, we contribute to the body of evidence that PLDR should be considered as an option for patients with prior irradiation presenting with bulky recurrences when an oncological resection is not possible. We report on two patients with locally recurrent chondrosarcoma who were not surgical candidates and thus underwent re-irradiation with PLDR. The first patient received re-irradiation with a total dose of 66 Gy and had a robust response with a 5-year survival with remarkably little toxicities. Our report therefore provides evidence, albeit anecdotal, that PLDR may mitigate the late toxicity associated with high cumulative doses after re-irradiation. In the second patient, we did not deliver a high dose due to significant pre-existing late toxicity from the initial course of definitive radiotherapy. Although there was an initial clinical response with improvement in brachial plexopathy, she eventually developed in-field progression of the superficial aspect of the tumor that caused skin ulceration 8 months later which we hypothesized was due to the skin-sparing effect of photons leading to under-dosing. She eventually succumbed to an unrelated condition.
Other novel techniques for delivering re-irradiation have been explored. These include spatially fractionated radiotherapy with either microbeams or specialized lead collimators; particle therapy with protons or carbon ions; or stereotactic radiotherapy. Each of these techniques exploits different radiobiological or dose-distribution mechanisms to improve tumor cell killing while minimizing normal organ toxicities. Spatially fractionated radiotherapy creates radiation low dose troughs and is hypothesized to improve immunological upregulation. Unfortunately, this technique requires additional resources in the form of training and equipment, and expertise may not be readily available. Particle therapy has a benefit over conventional radiotherapy through reduced low and intermediate-dose splash because of the inherent lack of an exit beam. Particle therapy is an attractive option for re-irradiation but is not always available within treatment networks; logistic and financial cost issues can also be prohibitive. Stereotactic body radiotherapy utilizes steep dose gradients to deliver high ablative doses and is an attractive option for small tumors. However, there can be significant toxicity for larger tumors, which may limit its utility for bulky recurrences. Compared to these modalities, PLDR is technologically easy to implement and requires no significant expense in equipment or training.
CNS tumors have been the focus of several PLDR re-irradiation studies [
8,
10-
12]. In a study of 103 patients with recurrent gliomas, Adkison et al. [
8] reported a median survival of 11.4 months for low-grade, 5.6 months for grade 3, and 5.1 months for grade 4 tumors. The median PLDR re-irradiation dose was 50 Gy and the average cumulative dose was 106.8 Gy. Patients with grade 4 disease who had a longer disease-free interval (<14 months vs. ≥14 months) before PLDR re-irradiation were found to have a longer median survival of 21 weeks versus 28 weeks, respectively. The mean treatment volume was 403 cm
3. Four patients were found to have necrosis on autopsy. However, only 15 of the 103 patients underwent autopsy and the true rate is likely higher. The authors concluded that PLDR is safe and allows for the retreatment of larger volumes [
8]. Murphy et al. [
10] reported the outcomes of 24 patients with a variety of CNS tumors who received PLDR re-irradiation for recurrent disease. The median dose for PLDR re-irradiation was 54 Gy, while the median target volume was 369.1 cm
3. With a median follow-up of 5.2 months, the median overall survival (OS) and 6-month OS after PLDR treatment was 8.7 months and 71%, respectively. The rate of grade 3 toxicity after PLDR re-irradiation was 18.1%, which was similar to the rate of 20.7% after initial therapy. There was no recorded event of necrosis, which is reassuring, considering that the group was heavily pretreated, with 50% or more having undergone 4 or more lines of systemic therapy. Bovi et al. [
12] investigated the addition of PLDR re-irradiation to bevacizumab in patients with recurrent high-grade gliomas. In this retrospective study of 80 patients, 47 patients were treated with bevacizumab alone, while 33 received additional PLDR re-irradiation. There were statistically significant improvements to the progression-free survival (PFS; 12 months vs. 4 months) and OS (16 months vs. 9 months) in the PLDR group. In a study of recurrent ependymoma (ages 3–31, with 8 lesions; 2 brains and 6 spinal cord) requiring large field re-irradiation post initial resection and adjuvant radiotherapy, patients received a median cumulative dose of 105 Gy after a median interval between radiotherapy of 58 months. The 4-year OS rate was 60% and no patients developed necrosis on serial magnetic resonance imaging scans, although one patient developed radiculopathy [
11]. The median portal volume for patients treated with 2D-planning techniques was 348 cm
2, while the median volume encompassed by the prescribed dose was 82.9 cm
3 for those treated with 3DCRT. With a notable median follow-up of 64 months, the estimated 4-year OS and PFS were 60% and 35.7%, respectively. In a recent publication, Kutuk et al. [
13] reported on their experience with PLDR re-irradiation in 18 patients with recurrent primary CNS malignancies. At a median follow-up of 8.7 months, the median PFS and OS were 5.7 months and 6.7 months, respectively. The median cumulative prescription dose was 102.7 Gy and symptomatic radiation necrosis occurred in three patients. The conclusion was that despite the high-cumulative dose to organs at risk, the tumor control and toxicity outcomes were within expectations. These studies suggest that in patients with CNS recurrences who have undergone previous radiotherapy, PLDR is an option that warrants consideration, especially when large-volume treatment is required. This is particularly useful when other options such as surgical resection or stereotactic radiosurgery are not feasible.
Richards et al. [
14] reported outcomes of 17 patients with breast cancer re-irradiation with PLDR with a median prior radiation dose of 60 Gy and median PLDR dose of 54 Gy. All patients exhibited a response—15 patients had a complete response, and two had a partial response. After a median follow-up of 18 months, two failed in-field. The estimated 2-year local control rate was 92%. Four patients had grade 3 acute skin toxicity. The median cumulative dose was 110 Gy. Two patients developed non-healing ulcers as late toxicity; both had complicated surgical histories with wound healing issues before PLDR. This outcome measures favorably against other data from conventional re-irradiation.
Limited data exists for PLDR in head and neck cancers. A case report described a patient with recurrent nasopharyngeal carcinoma who received PLDR as a third course of radiotherapy resulting in a total of 190 Gy delivered cumulatively [
15]. A complete response was achieved. However, the long-term follow-up outcomes of this patient were not described.
For gastrointestinal cancers, Yang et al. [
16] published a phase 2 study of 14 patients who received 50–54 Gy abdominal PLDR. Eleven of them exhibited partial responses, and three demonstrated disease stability. No serious adverse effects were observed. A later study by the same authors [
17] demonstrated that the combination of capecitabine and oxaliplatin, immunotherapy, and PLDR for gastric cancers with peritoneal dissemination was tolerable and efficacious with an objective response rate of 70.8%.
There is a paucity of data for sarcomas. PLDR for chondrosarcoma was briefly reported in a case series by Yan et al. [
18] in which a middle-aged patient with locally recurrent indolent chondrosarcoma received 54 Gy/27 fractions palliative re-irradiation after initial 50 Gy and the conclusion was that PLDR was safe and effective. Our case reports contribute to the scant literature on PLDR for sarcomas.
Lee et al. [
19] reported the outcomes of 39 patients who received PLDR re-irradiation using forward-planning techniques to targets in the thorax, abdomen, and pelvic region. With a median dose of 50.4 Gy and 50 Gy for first and re-irradiation and a median follow-up of 8 months, 79% experienced symptomatic improvement. Although 23% of patients experienced grade 2 or more toxicities, none developed grade 4 or 5 toxicities. In a study of 13 patients with recurrent cancer (head and neck, lung, and pelvis sites), Tong et al. [
20] reported responses in all patients—partial and complete responses in 10 and three patients, respectively. The previous dose received was ≥50 Gy in all patients, while the re-irradiation dose ranged from 10 Gy to 60 Gy. With a median follow-up of 27 months, all toxicities were grade 2 or lower, with only one patient developing grade 3 toxicity in the form of skin fibrosis.
In conclusion, PLDR is a novel radiotherapy technique that exploits the radiobiological phenomenon of low-dose hyper-radiosensitivity to maximize tumor killing in tumors while minimizing normal organ toxicities by encouraging sublethal repair. Compared to alternative re-irradiation modalities, PLDR is relatively simple to implement and can be used for large, bulky tumors. However, the low dose rate requires a longer time on couch. Our two cases highlight our early experience with PLDR irradiation for bulky recurrences of recurrent bony sarcoma. Notably, one patient has remained in long-term remission for 5 years without significant treatment sequelae. We also conducted a literature review of PLDR in re-irradiation and our findings recapitulate the safety and efficacy of PLDR as an option in the setting of re-irradiation of recurrent tumors where resection is not feasible. Although the data is encouraging, more prospective studies are required, particularly to compare its efficacy against conventional photons.