Short-course versus long-course neoadjuvant chemoradiotherapy in patients with rectal cancer: preliminary results of a randomized controlled trial
Article information
Abstract
Purpose
Colorectal cancer is becoming an increasing concern in the middle-aged population of Iran. This study aimed to compare the preliminary results of short-course and long-course neoadjuvant chemoradiotherapy treatment for rectal cancer patients.
Materials and Methods
In this clinical trial we recruited patients with rectal adenocarcinoma located from 5 cm to 15 cm above the anal verge. Patients in group I (short-course) received three-dimensional conformational radiotherapy with a dose of 25 Gy/5 fractions in 1 week plus concurrent XELOX regimen (capecitabine 625 mg/m2 from day 1–5 twice daily and oxaliplatin 50 mg/m2 on day 1 once daily). Patients in group II (long-course) received a total dose of 50–50.4 Gy/25–28 fractions for 5 to 5.5 weeks plus capecitabine 825 mg/m2 twice daily. Both groups underwent consolidation chemotherapy followed by delayed surgery at least 8 weeks after radiotherapy completion. The pathological response was assessed with tumor regression grade.
Results
In this preliminary report on complications and pathological response, 66 patients were randomized into two study groups. Mean duration of radiotherapy in groups I and II was 5 ± 1 days (range, 5 to 8 days) and 38 ± 6 days (range, 30 to 58 days). The median follow-up was 18 months. Pathological complete response was achieved in 32.3% and 23.1% of patients in the short-course and long-course groups, respectively (p = 0.558). Overall, acute grade 3 or higher treatment-related toxicities occurred in 24.2% and 22.2% of patients in group I and II, respectively (p = 0.551). No acute grade 4 or 5 adverse events were observed in either group except one grade 4 hematologic toxicity that was seen in group II. Within one month of surgery, no significant difference was seen regarding grade ≥3 postoperative complications (p = 0.333).
Conclusion
For patients with rectal cancer located at least 5 cm above the anal verge, short-course radiotherapy with concurrent and consolidation chemotherapy and delayed surgery is not different in terms of acute toxicity, postoperative morbidity, complete resection, and pathological response compared to long-course chemoradiotherapy.
Introduction
Colorectal cancer is the third most common cancer in males and the fourth most common among females in Iran [1]. The incidence of rectal cancer in our country is relatively lower than that of Western countries; however, it has been increasing rapidly in recent years. According to a study by Malekzadeh et al. [2], the incidence of colorectal cancer has increased by 80% in the last 30 years in Iran. Interestingly, up to the age of 45 years, the incidence of this cancer in the Iranian population does not significantly differ compared with the US population. Therefore, colorectal cancer is becoming a serious problem for the young Iranian population and the workforce [3].
Currently, the routine treatment plan for locally advanced rectal cancers comprises of preoperative radiation with or without chemotherapy followed by surgery. To date, two types of neoadjuvant therapy have been introduced for rectal cancer. The first method, known as long-course chemoradiotherapy (LCRT) or conventional chemoradiotherapy, includes 45–54 Gy in 25–28 fractions along with concomitant chemotherapy—mainly 5-fluorouracil (5FU) or its derivatives—followed by delayed surgery 6–8 weeks later. This method is mostly applied in the United States and several European countries [4]. Another method is the Northern-European method (especially in Scandinavia and Poland), known as short-course radiotherapy (SCRT), which consists of 25 Gy in 5 fractions without concomitant chemotherapy followed by immediate surgery within 1 week after radiotherapy completion [5].
One of the disadvantages of conventional LCRT with concomitant chemotherapy is the prolongation of the treatment course and the interval between diagnosis and surgery. Moreover, LCRT is costly. Likewise, in our non-private center, patients experience long waiting times for LCRT, whereas the time spent for SCRT is one-fifth of that in LCRT.
In most studies, SCRT without chemotherapy has shown lower pathological response rates compared with conventional LCRT [6], and the addition of chemotherapy to SCRT has always been associated with a concern about increased complications [7]. In addition, no theoretical consensus currently exists on a SCRT regimen that will yield the highest response rate [8,9].
According to our previous studies on short-course and long-course treatment in rectal cancer patients, and the promising results achieved with SCRT, we aimed to compare these two methods in terms of safety profile, pathologic response, and survival in a randomized controlled trial [10,11]. Here, we report the preliminary results of this study by 50% of the expected accrual, including treatment complications and pathological complete response (pCR). In the future, we will report late toxicities and survival rates.
Materials and Methods
This study was a randomized controlled clinical trial conducted at the radiation oncology ward of Cancer Institute of Iran. Patient recruitment began in April 2016. Patients with a confirmed histological diagnosis of rectal adenocarcinoma located within 5 to 15 cm from the anal verge, and cT3-4 stage or node positive status—based on magnetic resonance imaging (MRI) or endoscopic ultrasound (EUS)—were enrolled in the study. We excluded patients with distant metastasis, an Eastern Cooperative Oncology Group performance score >1, non-operable status or intolerance to chemotherapy, a history of current or past second malignancy, recurrence after previous surgery, and cases of familial adenomatous polyposis (FAP).
The pre-treatment evaluation consisted of imaging modalities such as pelvic MRI, EUS, and thoracoabdominal computed tomography (CT) scan and lab tests including complete blood count, liver and renal function tests, and serum carcinoembryonic antigen (CEA). After completion of staging workup examinations, patients who met the eligibility criteria were given informed consents. Patients who were willing to take part in the study were then randomly assigned either to the short-course or long-course treatment group. Patients of each group were matched in terms of stage of cancer. Randomization was based on permuted block method. Due to the nature of the study intervention, blinding of participants to the assignment group was not possible. In order to minimize patient loss and withdrawal, the investigators followed participants by telephone. After initiation of the study, physical examination and lab tests were performed weekly to assess post-treatment complications. The treatment regimen of group I (short-course) consisted of three-dimensional conformational radiotherapy (3D-CRT) with a total dose of 25 Gy in 5 fractions in 1 week plus concurrent XELOX (capecitabine 625 mg/m2 twice daily from day 1 to 5 and oxaliplatin 50 mg/m2 intravenous injection day 1 only). As for group II (long-course), patients underwent 3D-CRT with a total dose of 50–50.4 Gy in 25–28 fractions during 5 to 5.5 weeks plus concurrent capecitabine 825 mg/m2 twice daily. Capecitabine tablets were provided by the Actero Middle East Company (a.k.a. Actero Pharma in Tehran, Iran) for all participants. Patients of both groups underwent delayed surgery 8 weeks after the completion of radiotherapy which was performed in either the surgical oncology ward or the colorectal surgery ward. They also received pre-operation chemotherapy with XELOX 3 to 4 weeks after radiotherapy completion.
1. Outcome assessment
The primary outcome of interest was acute toxicity during chemoradiotherapy up to 1 month of its completion based on the Common Terminology Criteria for Adverse Events (CTCAE) v3.0. In the present report, the secondary outcomes were pCR and down-staging, complete resection, and post-operative morbidity. Pathological response was defined by tumor regression grade (TRG). We used the modified Ryan system that had been endorsed by the American Joint Committee on Cancer [12]. Overall down-staging was defined as conversion of clinical stage to ypT0-2N0, which is considered as non-locally advanced disease. Tumor down-staging was defined as the conversion of primary tumor to ypT0-2. Nodal down-staging was characterized only among those with cN1-2 if ypN was less advanced than cN. Post-operative morbidity was characterized as any toxicity attributable to surgery up to 1 month after the procedure.
2. Treatment planning
Contouring of clinical target volume (CTV) was based on the Radiation Therapy Oncology Group consensus [13]. Delineation of lymph node basins at risk was based on the international guideline by Valentini et al. [14]. Planning target volume (PTV) was generated by planning software and with adding an 8-mm margin in all dimensions. Before initiation of treatment, the definite treatment plan was approved by the patients’ physician regarding 95% dose coverage of PTV, dose and location of Dmax, and dose to organs-at-risk.
3. Statistical analysis
Sample size was calculated according to a previous study performed at this center and another study in which the reported incidence of grade ≥2 toxicity was 50% and 75% in SCRT and LCRT, respectively [10,15]. The power was 80% and type I error (α) was 0.05. Taking into account a 10% loss, the required sample size was calculated as 120 patients (60 in each group). Since this was a preliminary analysis, we included 50% of the total expected accrual size. We used the following formula to calculate sample size [16]:
n = (Zα/2 + Zβ)2 × (p1(1–p1) + p2(1–p2)) / (p1–p2)2
For evaluating pCR and surgical and chemoradiotherapy complications, the chi-square test and multivariate logistic regression were used. A p-value of less than 0.05 was considered as statistically significant.
4. Ethical considerations
Informed consent was obtained from all participants prior to enrollment in the study. This study was approved by the Ethics Committee of Tehran University of Medical Sciences and the Iranian Registry of Clinical Trials (Ethics Code: IR.TUMS.VCR.REC.1396.3475, IRCTID: IRCT2017110424266N3).
Results
Initially, 123 patients were recruited; however, after consideration of inclusion and exclusion criteria, only 66 patients were allocated to receive either SCRT (group I) or LCRT (group II). No patient was lost to follow-up or discontinued intervention (Fig. 1).
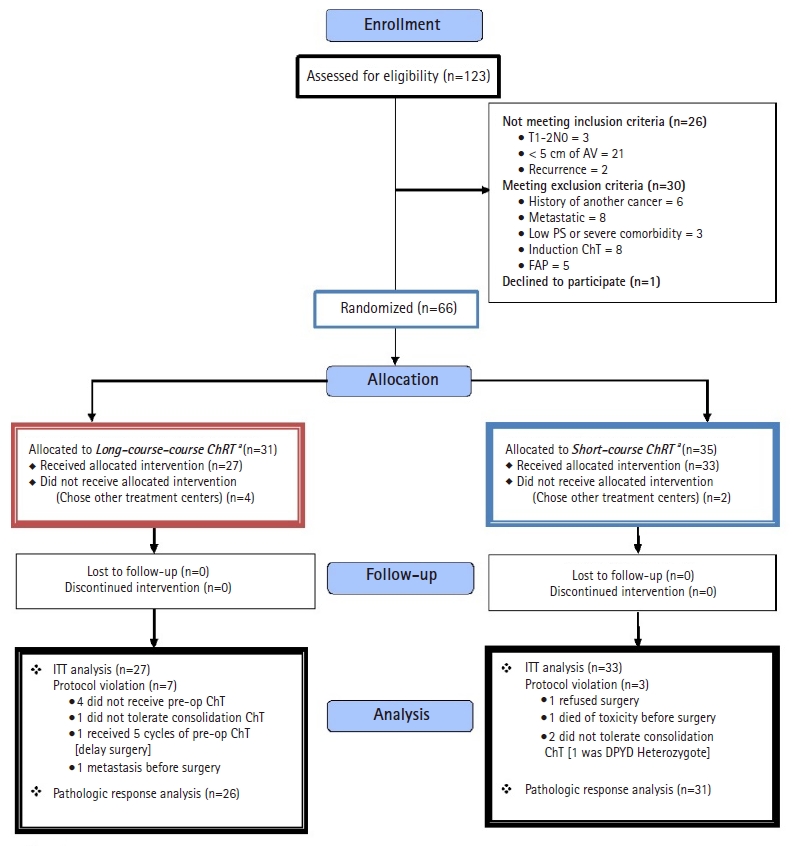
CONSORT flow diagram of the study. AV, anal verge; PS, performance score; ChT, chemoradiotherapy; FAP, familial adenomatous polyposis; ITT, intention-to-treat.
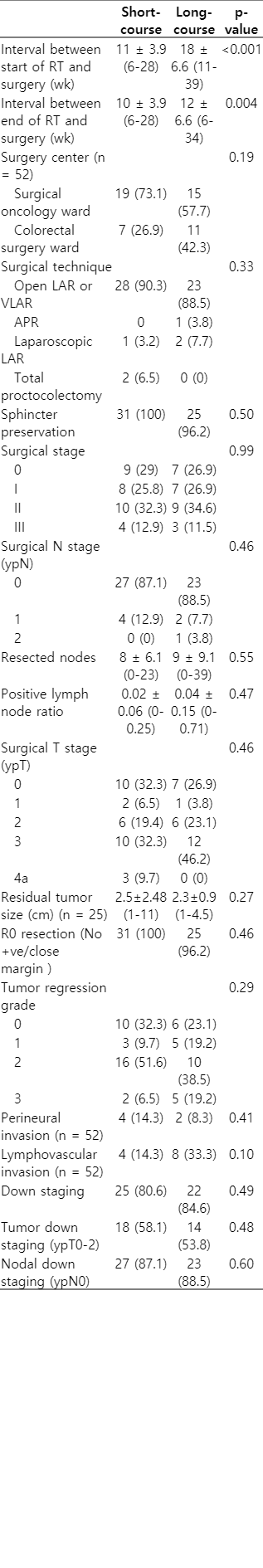
1. Baseline characteristics
The baseline characteristics of patients in both groups are demonstrated in Table 1. As shown, there was no significant difference between the two groups in respect to the studied variables. The median age of patients in the short-course and long-course treatment groups was 56 ± 10.3 and 53 ± 12.9 years old, respectively. In both groups, the majority of patients had a histologic grade 1 tumor.
2. Acute treatment toxicity
The mean duration of radiotherapy course in the SCRT and LCRT groups was 5 ± 1 and 38 ± 6 days, respectively. Grade 2 and higher acute adverse events (AEs) were observed in 75.8% and 61.5% of patients in group I and II, respectively (p = 0.19). The percentage for grade 3 and higher AEs in the concurrent chemoradiotherapy period was 15.2% and 14.8%, respectively (p = 0.63). No grade 4 or 5 radiotherapy-induced adverse event was observed except one grade 4 hematologic toxicity in the LCRT group.
Grade 3 and higher AEs related to preoperative chemotherapy (consolidation) were observed in 12.1% and 11.5% of patients in group I and II, respectively (p = 0.64). Preoperative chemotherapy tolerance (receiving full planned dose) in group I and II was 87.9% and 81.8%, respectively (p = 0.4). Collectively, grade 3 or higher acute treatment toxicities acute treatment toxicities, including chemoradiotherapy- and consolidation chemotherapy-attributable AEs, were seen in 24.2% and 22.2% of patients in group I and II, respectively (p = 0.55).
The frequency of the most severe acute toxicities is demonstrated in each treatment group (Table 2).
3. Surgical outcomes
Fifty-five percent of the surgical specimens were re-examined by a skilled pathologist. TRG was altered in 6/33 (18.2%) cases; of note, however, only one change from grade 1 to grade 0 was observed, and the majority of changes were between grades 2 and 3. A pCR (final TRG = 0) in the SCRT and LCRT groups was achieved in 32.2% and 23.1% of cases, respectively (p = 0.56); whereas a pathological complete or near complete response (final TRG = 0–1) was seen in 41.9% and 42.3% of cases, respectively (p = 0.99). One patient had ypT0N1 that was considered TRG = 0, however, his/her surgical stage was classified as stage 2. Tumor down-staging (ypT0-2 ypN0, based on the definition) occurred in 54.8% and 53.8% of the cases in group I and II, respectively (p = 0.58). Detailed data is shown in Table 3.
4. Post-operative morbidity
The frequency of grade 3 or higher post-operative morbidities (within 1 month after surgery) in the SCRT and LCRT groups was 19.4% and 11%, respectively (p = 0.33). No post-operative mortality was observed. The type of post-operative morbidities in each group is shown in Table 4.
5. Late treatment toxicity
The frequency of grade 2 or higher late treatment toxicities (at least 6 months after radiotherapy) in the SCRT and LCRT groups were 38.7% and 38.4%, respectively (p = 0.56); while grade 3 and higher toxicities were seen in 6.5% and 11.5% of the patients in group I and II, respectively (p = 0.16) (Table 5). The frequency of late treatment toxicities varied based on the ward in which surgeries were performed (Table 6); in the colorectal surgery ward, toxicities were less frequently observed in patients who received the short-course treatment compared with patients who received long-course treatment (p = 0.02).

Late treatment toxicities and frequency of the most severe complications in SCRT (group I) and LCRT (group II)
Discussion and Conclusion
We evaluated the preliminary outcomes of SCRT with concomitant XELOX regimen in comparison to LCRT with concomitant capecitabine. Both study groups received consolidation chemotherapy with XELOX at the resting interval before surgery. The reason for administering XELOX in the concurrent regimen of patients undergoing SCRT was the promising results we achieved in the previous single-arm study conducted at our institution [10]. Also, another study published a few years ago showed that the addition of oxaliplatin leads to more robust down-staging compared with capecitabine alone [17]. Considering these results, we aimed to investigate the efficacy and safety of XELOX regimen for concurrent chemotherapy in SCRT as a potential alternative option compared with the standard treatment, which is LCRT with fluorouracil or capecitabine. As for the consolidation regimen, we used the adjuvant therapy for locally advanced rectal cancer, which is primarily XELOX, since oxaliplatin has been reported to enhance disease-free survival compared with capecitabine alone or fluorouracil plus leucovorin alone [18,19].
Some patients experienced a relatively long interval from radiation completion to surgery. These outliers were either due to temporary loss to follow-up, long waiting lists of the surgical wards, or occasional lack of coordination between the departments. Although none of the patients experienced re-growth of the tumor at the time of surgery, this issue should be taken into consideration since interval prolongation might affect pathological response.
In this study, there was no significant difference in respect to acute treatment toxicities and post-operative morbidities between the short-course and long-course treatment groups. In LCRT, the peak of radiotherapy adverse events, including proctitis, enteritis, diarrhea, and mucositis is within the third to fifth week of treatment and the severity of the acute toxicities will gradually decrease 3 to 4 weeks after the completion of therapy. In this group, the patient receives chemotherapy simultaneously along with radiotherapy at the time when toxicities are at their peak, which can affect the severity of toxicities [20]. It is assumed that more acute toxicities are faced in the short-course treatment which uses the hypofractionated regimen, as it is a kind of accelerated regimen. According to a previous Polish study, in SCRT, acute toxicities peak at the 11th to 14th day after radiotherapy. At this time, the patient is resting after a 5-day treatment course, so he/she is not exposed to chemotherapy or radiotherapy; on the other hand, delaying the surgery for at least 8 weeks after irradiation will reduce the severity of toxicities [21]. In SCRT, malignancy symptoms (obstruction, bleeding, rectal discomfort) resolve sooner due to larger fractions and thus, influence the decline in the feeling of discomfort from acute toxicities. The lack of statistically significant difference in acute toxicities between the two groups can be explained by the mentioned reasons.
Another interesting finding of this study was that despite a smaller equieffective dose of radiotherapy (EQD2 = 31.25 Gy vs. 50 Gy) and also a smaller total chemotherapy dose (5 days vs. 25 days of capecitabine delivery) in the SCRT group, short-term oncologic outcomes were the same in both groups. More interestingly, in the SCRT group, the pCR was higher than that in most of the previous similar studies [22-27]. This finding can be rationalized by the fact that the large fractions used in a hypofractionated radiotherapy regimen can induce an immune response that will eventually increase the biologic effects of concomitant and consolidation chemotherapy. This immune response results from the release of a great number of antigens due to the breakdown of tumoral cells, and the presentation of these antigens to T cells [28,29]. Nevertheless, this hypothesis needs further assessment.
Comparison of our study with other similarly designed studies in which chemotherapy is applied along with SCRT concluded that we had reached more favorable outcomes (Table 7). So, the question is why short-course chemoradiation has produced such a good result. There are several reasons that could explain the more favorable results in this study; firstly, the precise delineation of target volumes based on international guidelines [14] and the strict confirmation of treatment plans, considering the sufficient coverage of PTV and also the dose of organs-at-risk; second, administration of capecitabine with radiotherapy instead of bolus 5FU is shown to be associated with fewer toxicities and higher response rates in a study by Haddad et al. [30]; third, prolonging the interval between radiotherapy completion and surgery to more than 8 weeks, as this has been demonstrated by Rega et al. [31] to reduce adverse events and increase response to neoadjuvant therapy. In our study, the interval from end of radiotherapy to surgery was higher compared with the majority of other similar studies. Consistent with our trial, a study by Myerson et al. [26], with a similar interval of 17 weeks, reported pathological responses comparable to our results; moreover, delivering consolidation chemotherapy before surgery which has been shown to be associated with an increase in complete response rates in a study by Habr-Gama et al. [32]; addition of oxaliplatin to capecitabine simultaneously with radiotherapy, which despite controversies, has been proved to increase response rates [17]; and lastly, performing operations in a more specialized ward for colorectal surgery by skilled and experienced colorectal surgeons.
In conclusion, SCRT versus LCRT with consolidation chemotherapy and delayed surgery were not significantly different in regards to acute toxicities, post-operative morbidities, complete resection, and pathological response. However, we should wait longer to be able to make a definitive comment on local recurrence, distant recurrence, survival rates, and late toxicities. Since our patients were only followed for a relatively short period of 18 months, results on these long-term measures will be reported separately in future studies. Prospective studies should focus on using novel radiotherapy techniques for reducing grade 2 acute and late AEs. Moreover, comparison of SCRT with sequential versus simultaneous chemotherapy, investigation of tumor and mesorectum dose-escalation with SCRT to increase pCR rates, and implementation of a watch-and-wait approach after SCRT + chemotherapy is suggested. Conclusively, re-performing the current study with removal of simultaneous oxaliplatin, and based on the substantial pCR, and investigating sphincter preservation in lower rectal tumors (less than 5 cm from the anal verge) is recommended.
Notes
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Acknowledgements
We thank the Actero Middle East Company (a.k.a. Actero Pharma) providing the capecitabine tablets for both groups.
This study was funded by the Vice-chancellor of Research, Tehran University of Medical Sciences (Grant ID: 95-04-207-33683).