 |
 |
AbstractPurposeAlthough the conventional gamma ray brachytherapy has been successful in treating endometrioid endometrial adenocarcinoma (EC), the molecular and cellular mechanisms of this anti-tumorigenic response remain unclear. Therefore, we investigated whether gamma ray irradiation induces changes in the number of FoxP3+ T-regulatory lymphocytes (Tregs), CD56+ natural killer cells (NK), and the expression of progesterone receptor membrane component 1 (PGRMC1) in the tumor microenvironment (TME).
Materials and MethodsAccording to the inclusion criteria, 127 cases were selected and grouped into irradiation-treated (Rad+) and control (underwent surgery) groups and analyzed using immunohistochemistry. Predictive prognostic values were analyzed using Mann-Whitney U test, ROC analysis, relative risk, log-rank, Spearman rank tests and multivariate Cox’s regression.
ResultsWe observed significant differences (p < 0.001) between the radiation-treated patients and the control groups in FoxP3+ Tregs numbers, CD56+ NK cells and PGRMC1 expression. Gamma ray induced a 3.71- and 3.39-fold increase in the infiltration of FoxP3+ cells, CD56+ NK cells, respectively and 0.0034-fold change in PGRMC1 expression. Univariate and multivariate analyses revealed predictive role of the parameters. In the irradiated patients’ group, inverted correlations between clinical unfavorable outcome, FoxP3+ Tregs and CD56+ NK cells were observed.
IntroductionThe most common type of endometrial cancer is endometrioid endometrial carcinoma (EC), an estrogen-dependent neoplasm that accounts for 80%–85% of all cases, including the non-estrogen-dependent tumors [1]. The EC is frequently associated with obesity, diabetes, hypertension, nulliparity, early menarche, and late menopause which are known to increase plasma levels of estrogen and decrease in progesterone levels [1-4]. Due to the altered physiology, the tumor microenvironment (TME) plays a major role in promoting survival and progression of the growing tumor. The TME consists of extracellular matrix, stromal cells such as fibroblasts, mesenchymal stromal cells, pericytes, occasional adipocytes, blood and lymphatic vascular networks, and immune cells [5]. This complex crosstalk between these cellular and molecular pathways, particularly the immune cells, within the TME creates obstacles to effective treatments. However, radiation therapy has shown promise in penetrating the solid TME and remains the gold standard of endometrial cancer treatment [6]. With increasing knowledge of the role of tumor-infiltrated immune cells and hormonal signaling, interest in the mechanisms and the effect of brachytherapy on the TME is growing [7].
Forkhead box P3 (FoxP3) is a key transcription factor in the development and function of T regulatory cells (Tregs) that is considered to be the most specific Tregs marker to date [8]. In physiology, FoxP3+ Tregs suppress antitumor responses and thereby, maintain immunological tolerance to host tissues [9]. Shang et al. reported that high FoxP3+ Tregs infiltration was associated with significantly longer cancer-specific survival in endometrial cancer [10]. However, there is controversial data about the role of FoxP3 cells in prognosis in endometrial cancer. For instance, FoxP3+ T-lymphocytes were shown to be associated with decreased survival [11].
Another group of tumor-infiltrated immune cells, natural killer (NK) lymphocytes, belong to the innate immune system which acts by lysing cells lacking major-histocompatibility-complex (MHC) proteins and secreting cytokines [12]. Although the majority of NK cells are found in peripheral blood, spleen and bone marrow, they can also reside in lymph nodes, gut, liver and uterus [13], and have been shown to be impacted by radiotherapy. The indirect effects of radiotherapy on NK cells can be divided into three categories: the modulation of activating and inhibitory NK ligands, the release of damage-associated molecular patterns (DAMPs), and the enhancement of NK cell migration to the tumor. Multiple irradiated cancer cell lines showed an increased expression of the intracellular adhesion molecule 1 (ICAM1), which was described to enhance NK cell-mediated killing by increasing cell-to-cell adhesion, and the Fas receptor, possibly indicating higher susceptibility to NK cell-mediated apoptosis [14]. NK cells directly interact with tumor cells and also influence the function of other innate and adaptive immune cells in the TME [15]. Experimental data of irradiation of co-cultivated human blood NK cells and various cancer cell lines revealed that NK cells sensitize tumor cells to radiation and radiation sensitizes tumor cells to NK cell attack, resulting in an increased tumor cell killing [16].
EC is a hormone-dependent disease where the expression of progesterone receptors has been associated with histological tumor differentiation, response to radiotherapy and metastatic potential [17]. However, the same non-canonical progesterone receptors such as progesterone receptor membrane component 1 (PGRMC1) shows immunosuppressive effect at the TME of breast and hepatocellular carcinoma [18,19]. PGRMC1 is a heme-containing protein that interacts with epidermal growth factor receptor (EGFR) and cytochromes P450 in cancer cells [20]. PGRMC1 is induced in different types of cancers such as tumors of the lung, thyroid and colon [21] and plays a causative role in cancer progression. For instance, in vitro, PGRMC1 induces tumor cell proliferation, chemotherapy resistance and invasion, and in vivo, it increases tumor growth, induces angiogenesis and aids metastasis [22].
Although no report is available describing possible mechanisms between these cells, tumor infiltrated FoxP3+ Tregs, NK cells and PGRMC1 play important roles in and may potentially contribute to the remodeling of the TME post-intracavitary gamma-ray brachytherapy by modulating immune response to cancer and inhibiting signaling pathways. Thus, we investigate whether gamma ray irradiation leads to the modulation of the TME in EC by inducing immunogenic changes in NK, FoxP3 cells infiltration and PGRMC1 expression. Additionally, we examine a potential association between expressions of these markers. Moreover, we suggest that such parameters of TME could be potential clinical biomarkers for ECs that are resistant to gamma-ray brachytherapy irradiation and prognostic factor for disease-free survival (DFS).
Materials and Methods1. PatientsThis prospective study involved women with EC who were treated between 2014 and 2017 in two clinical oncological centers in Belarus. We considered the following as inclusion criteria: a presence of EC, clinical stage I–III (by the International Federation of Gynecology and Obstetrics [FIGO], 2009), an absence of malignant tumors of other localizations during life, an absence of autoimmune diseases and immunodeficiency syndromes. The exclusion criteria were non-endometrioid carcinoma, clinical IV stage, a presence of synchronous and metachronous malignancies, presence of autoimmune diseases and immunodeficiency syndromes. According to our inclusion criteria, a total of 127 EC out of 617 cases of endometrial carcinomas were eligible for the study (Fig. 1).
All patients were divided into two groups: patients who received preoperative gamma-ray irradiation (Rad+ group, 59 cases) and patients who underwent surgical procedures only (control group, 68 cases). Preoperative intracavitary brachytherapy in Rad+ group was performed 48 hours before hysterectomy. The 60Co source was placed in the uterine cavity and a total boost dose of 13.5 Gy gamma-ray irradiation was administered in these patients. All patients included in this research received treatment according to the Ministry of Healthcare Protocols of Diagnostics and Treatment of Oncological Diseases (Republic of Belarus). Patients were consented in this study and an ethical approval was obtained and reviewed by the Institutional Review Board, Gomel, Belarus (No. 15.05.2020). The clinical characteristics of patients are presented in Table 1.
2. ImmunohistochemistrySections (3–4 µm) were prepared from paraffin blocks and mounted on Thermo Super-Frost poly-L-lysine coated slides (Thermo Fisher Scientific GmbH, Schwerte, Germany). The slides were placed in Tris pH 9.0 retrieval buffer. Antigen retrieval was performed using a microwave oven, without boiling the buffer solution. After cooling of the slides at room temperature, endogenous peroxidase was blocked with 5% hydrogen peroxide. A 5% goat serum was used for blocking of the nonspecific antibody binding. Sections were washed by phosphate buffer solution (pH 7.4) after every step of staining. The following primary antibodies were added: FoxP3 (1:100; Abcam, Bristol, UK), CD56 (ready-to-use; Diagnostic Biosystems, URM) and PGRMC1 (1:150; Abcam) and the slides were incubated in a moist chamber at room temperature. Next, these slides were rinsed in wash buffer and incubated with secondary anti-mouse/rabbit antibodies. The visualization of product reaction was performed using 3,3-diaminobenzidine (DAB) chromogen, followed by Mayer’s hematoxylin counterstaining [23].
3. Morphometrical analysisThe morphometrical analysis was carried out using ImageJ (https://imagej.nih.gov/ij/) and HumaScope Premium LED (HUMAN Diagnostic, Wiesbaden, Germany) in 10 non-overlapping ×400 magnification power fields of area of cancer invasion in stroma. The number of FoxP3+ cells after count was inverted into number of cells/mm2 by formula:
where, FoxP3 is number of FoxP3+ cells in 1 mm2;
Number of CD56+ cells after count were inverted in number of cells/mm2 by formula:
where, NK is number of CD56+ cells in 1 mm2;
Expression of PGRMC1 was presented as number of positive cells to total number of the cells in 10 non-overlapping HPFs.
4. Statistical analysisThe power analysis (power = 80%, α = 0.05) from our pilot study (unpublished) on three cases with preoperative irradiation and five without in five HPFs, revealed that the minimum number of subjects is 58. Data was presented by the median lower and upper quartiles. Mann-Whitney U test was used for comparing of the study groups. ROC analysis was used to evaluate the threshold criterion, including area under the curve (AUC). A relative risk (RR) analysis was performed using Fisher two-tail test. According to the threshold criteria, the log-rank test and multivariate Cox proportional hazards regression analyses were employed for Rad+ group to determine differences in DFS and hazard ratio (HR). Next, correlations between groups of cases were assessed using a Spearman correlation test. All data analyses and visualization were performed using GraphPad Prism v.8.4.3 (GraphPad Software Inc., San Diego, CA, USA) and R Software v4.0.5 (R Foundation for Statistical Computing, Vienna, Austria) and packages dplyr 1.0.5, corrplot 0.89 and Hmisc 4.5-0.
Results1. FoxP3+ Treg cellsIn the Rad+ group, FoxP3+ expression was significantly higher (p < 0.0001) in the stroma of the tumor near the glands (Fig. 2A) (median, 110.8 cells/mm2; range 55.4 to 213.5 cells/mm2) compared to the control group (median, 41.5 cells/mm2; range, 13.6 to 55.4 cells/mm2) (Figs. 2B, 3A). A threshold for the total number of Treg cells of 67.4 cells/mm2 was adopted as the best differentiating value between patients with or without gamma-ray preoperative brachytherapy, with 86.8% specificity and 69.5% sensitivity (AUC = 0.76; p < 0.0001). The gamma ray irradiation increased the likelihood of the presence of FoxP3+ cells by 3.71 times over the threshold criteria (RR = 3.71; 95% confidence interval [CI], 2.92 to 5.78; p < 0.0001). In DFS analysis of cases with a focus on FoxP3+ cells/mm2, it was found that the survival rate was statistically lower (p < 0.0001) in patients whose tumors had less than <67.6 cells/mm2 (HR = 6.199; 95% CI, 2.650 to 14.500), compared with the control group (Fig. 3B).
2. NK cellsCD56+ cells predominantly form medium density diffuse infiltrates in the in the cancer stroma (Fig. 2C) in the Rad+ group compared to the control group with fewer CD56+ cells infiltrating the EC glands (Fig. 2D). Median of CD56+ NK cells in the Rad+ group and the control group was 87.4 cells/mm2 (range, 55.4 to 138.5 cells/mm2) and -34.5 cells/mm2 (range, 13.8 to 55.4 cells/mm2), respectively. The number of CD56+ cells was significantly higher (p < 0.001) in Rad+ group compared to the control group (Fig. 3C) The AUC was 0.81 (p < 0.0001). The sensitivity, specificity, and threshold criteria were 67.8%, 91.2%, and 67.3 cells/mm2, respectively. The irradiation of the tumor increases the probability of infiltration of CD56+ cells which is 3.39 times higher than the threshold criteria (RR = 3.39; 95% CI, 2.27 to 5.61; p < 0.0001). The study of the DFS of EC cases showed that the survival rate was statistically lower (p = 0.0239) in patients whose number of CD56+ NK cells in tumors was fewer than 67.4 cells/mm2 (HR = 2.625; 95% CI, 1.136 to 6.060) (Fig. 3D).
3. PGRMC1 expressionOur microscopic analysis with Rad+ sections revealed that the intensity of PGRMC1 expression was weaker in the cancer stroma (Fig. 2E), whereas in the control group this expression was high in both glands and stroma (Fig. 2F). We observed a loss of PGRMC1 expression with a median 34.2% (range, 23.5% to 45.5%) in the Rad+ patients, compared with the control group -74.7% (range, 47.1% to 79.4%). PGRMC1 expressions were significantly lower (p < 0.001) in the Rad+ group compared to control (Fig. 3E). The AUC from ROC analysis of PGRMC1 expression was 0.85 (p < 0.0001). The sensitivity, specificity, and threshold criteria were 60.3%, 98.5%, and 67.8%, respectively. The PGRMC1 expression in the Rad+ group after gamma ray irradiation was 29.4 times lower than the threshold criteria (RR = 0.034; 95% CI, 0.006 to 0.187; p < 0.0001). The DFS was statistically lower (p < 0.0001) in patients whose PGRMC1 expression in the TME was more than 67.8% (HR = 0.027; 95% CI, 0.006 to 0.1154), as expected (Fig. 3F).
4. Multivariate analysisA multivariate Cox regression analysis revealed that all three parameters are independent prognostic factors of DFS of patients with EC (Table 2).
5. Correlation analysisWe differentially analyzed correlations between each group. In the control group (Fig. 4A), the clinical unfavorable outcome of the patients correlated with their age (p < 0.001; r = 0.61) and the FIGO (p < 0.001; r = 0.42). Additionally, a correlation was also revealed between the age of the patients and the FIGO stage (p < 0.001; r = 0.34) in this group (Fig. 4A). Furthermore, PGRMC1 expression positively correlated with EC grades (p < 0.001; r = 0.42) in the control group (Fig. 4A). In further analysis of the control group, the number of CD56 cells inversely correlated with the age of the patients (p < 0.001; r = -0.75) and clinical unfavorable outcome (p < 0.001; r = -0.59) (Fig. 4A). Additionally, an inverse correlation was revealed between the number of FoxP3 cells and PGRMC1 expression (p < 0.001; r = -0.69) (Fig. 4A).
In the Rad+ group (Fig. 4B), a positive correlation was observed between the age of the patients and clinical unfavorable outcome of EC (p < 0.001; r = 0.30). The number of CD56+ cells inversely correlated with patients’ age (p < 0.001; r = -0.37) and clinical unfavorable outcome of EC (p < 0.001; r = -0.39) (Fig. 4B). Additional correlation analysis revealed the following; an inverse correlation between the FIGO stage and the grade of the EC (p < 0.001; r = -0.41), and the number of FoxP3+ cells and clinical unfavorable outcome (p < 0.001; r = -0.71) in these patients (Fig. 4B).
Discussion and ConclusionA presence of immune cells in the TME is considered indicative of the host immune response against the growing cancer. Due to the potential anti-tumor role of immune cells, efforts have been in place to dissect and understand the various cellular cross-talks of tumor-immune infiltrates in the cancer stroma [24]. PGRMC1 is a protein with multiple functions such as prosurvival, anti-apoptosis, cell cycle progression, and TME modulation [25]. It was thought that radiation induced a suppressive effect on the immune system due to the radiosensitive nature of the tumor-infiltrated lymphocytes. The current understanding is that radiation can stimulate the immune system [26,27]. The present study investigates gamma ray irradiation-induced changes of host immunity and PGRMC1 expression in the zone of tumor invasion of the corpus uteri in EC.
According to Devaud et al. [28], FoxP3-expressing Tregs can be manifested in discrepant roles in cancer. For instance, they may suppress the effector immune response and help the tumor grow, whilst downregulating inflammation and thereby protecting the host from tumor progression [28]. Beauford et al. [29] revealed that human Tregs from healthy donors are more resistant to radiation-induced death than other immune cells. An increase in FoxP3 cell numbers in the TME is possibly induced by two mechanisms; post-radiation transforming growth factor-β1 (TGF-β1) secretion by cells in the cancer stroma and radioresistance of tumor-infiltrated Tregs. Irradiation is known to provoke an increase in the levels of TGF-β1, a cytokine involved in the Treg maturation [30]. An increased Tregs accumulation was observed in the tumor tissues after irradiation in bladder, breast and head and neck cancers [31,32]. Interestingly, we found an increase in the number of FoxP3+ cells in the EC TME compared to non-irradiated EC cancers. Furthermore, we demonstrate that gamma ray irradiation causes an increase in the probable FoxP3+ Treg cells infiltration that is over 3.71 times higher than the threshold criteria. This may indicate a role for irradiation-induced TGF-β which may lead to an increase in migration of FoxP3+ Tregs to the TME. Possible role of FoxP3+ cells of favorable DFS could be associated with the fact that FoxP3+ Tregs kill macrophages and monocytes and suppress their pro-tumorigenic effect. As a result, the positive impact of FoxP3+ Tregs may partially attribute to downregulate an unresolved inflammatory response which could promote tumor progression [33].
NK cells have been characterized as effector cells of the innate immune system with potent cytotoxic activity. As the first line of defense against tumor cells and viruses, NK lymphocytes are innately programmed to kill, without prior pre-activation, through patterned recognition of target ligands [34]. Tumor-infiltrating NK cells are associated with better DFS prognosis in patients with non-small cell lung cancer, clear cell renal cell carcinomas and colorectal cancer [35]. NK cells induce killing of tumor cells both by direct cytolytic activity and producing cytokines such as interferon-gamma (IFNγ) [36]. Tumor cells which were exposed to irradiation demonstrated an increased expression of the surface proteins MHC class I chain-related protein A and B (MIC-A/B) and human cytomegalovirus UL16-binding proteins (ULBP1-3), which are ligands for NK cell migration [37]. Yoon and colleagues revealed that irradiation in therapeutic doses generate large numbers of highly cytotoxic NK cells and the effective migration of expanded NK cells to target cancer cells [38]. In our study we show that gamma ray irradiation increases CD56+ NK cells infiltration of EC by 3.39 times higher than the threshold criteria. This was an interesting observation as the number of NK cells has been shown to be reduced in the TME of endometrial carcinoma [39].
PGRMC1 is a 195-residue protein which remains bound to cell membrane and contains a short luminal peptide, a single N-terminal transmembrane domain, and a C-terminal cytochrome b5-related heme-binding domain [40]. PGRMC1 is a non-classical progestin receptor [41]. This protein is found to be overexpressed to promote tumor growth in several cancers, including ovarian, endometrial, breast, lung and colorectal cancer [42]. Ahmed et al. [43] reported that PGRMC1 co-localizes with EGFR in cytoplasmic vesicles and regulates expression of EGFR. Interestingly, brachytherapy causes a downregulation of EGFR overexpression in cancer cells [44]. Our results show that gamma ray irradiation significantly decreases PGRMC1 expression by 29.4 times (compared to the threshold criteria) in EC, suggesting a co-inhibitory effect on both EGFR and PGRMC1 co-localization.
The results of univariate, multivariate survival and relative risk analyses suggest that an increased number of FoxP3+ and CD56+ cells and a reduction of PGRMC1 expression post-preoperative brachytherapy predict higher DFS of the patients with EC by modulating the tumor microenvironment. We show that the expression of FoxP3+ Tregs is higher in patients receiving radiation than the threshold criteria which was associated with longer DFS. Our data agrees with the data of Shang et al. [10] who showed that high FoxP3+ Tregs infiltration was associated with significantly longer cancer-specific survival in endometrial cancer (odds ratio = 0.42; 95% CI, 0.24 to 0.73, p = 0.002). Our study also showed that increased CD56+ NK cells infiltration predicts favorable prognosis of patients with EC, agreeing with a previous report where a high number of NK-lymphocytes was shown to predict better survival for solid tumors [45]. A low expression of PGRMC1 in our study correlated with favorable prognosis, which is an interesting observation as high PGRMC1 expression has been known to play a role in the activation of vascular endothelial growth factor involved in angiogenesis, tumor growth and metastasis [46].
Correlations between clinical parameters presented in our study are well known and were described before. In the control group, high grade tumors demonstrate more aggressive potential and a poor prognosis, so correlation between the grade of EC and PGRMC1 expression could be associated with it as has been shown in breast cancer by Ruan, and colleagues [47]. The number of FoxP3+ Tregs inversely correlate with PGRMC1; we suppose that this interaction is an indirect indication of the immune suppressive function of PGRMC1 during FoxP3+ Tregs infiltration in EC [47]. Not surprising that in both groups we observed an inverse correlation between the number of CD56+ NK cells and the age of the patients. However, this correlation was smaller in the Rad+ group, suggesting that gamma ray irradiation moderately increases the number of CD56+ NK cells in the cancer stroma possibly by increasing its migration to the TME [39,48].
Gamma radiation further induces changes in the TME of the EC in Rad+ group. For example, an inverse correlation is observed between the number of CD56+ NK cells and clinical unfavorable outcome of patients which indicates that gamma radiation enhances local immune response by increasing the number of CD56+ NK cells in the TME. A similar observation is found between the number of FoxP3+ cells and clinical unfavorable outcome, suggesting a good prognostic value of this type of Tregs in the survival of patients with EC.
In conclusion, this study presents changes in the number of FoxP3+, CD56+ cells, expression of PGRMC1 and their interactions in the EC TME after gamma ray irradiation. We also show that an association of this immunomodulation with disease free survival. The threshold criteria for these factors may have predictive values for clinical brachytherapy treatment and therefore, should be considered for the development of a combination therapy of brachytherapy and immunotherapy for EC. However, a large cohort study should be performed to further confirm our findings.
AcknowledgmentsWe would thank Veyalkin Ilya Vladimirovich for his consultation in statistical analysis. This study was support by initial grant of the Gomel State Medical University “Development and implementation of a prognostic model of the endometrioid adenocarcinoma of the uterine body based on pathological parameters of the tumor microenvironment,” registration number 20190038, registration date 24.01.2019, Gomel, Belarus.
Fig. 2.(A) Dense FoxP3+ cells (red arrows) infiltration of cancer stroma in irradiated group. (B) FoxP3+ Treg lymphocytes (red arrows) infiltrating stroma in area of invasion in control group. (C) A group of CD56+ NK cells (red arrows) in cancer glands in the irradiated group. (D) CD56+ NK lymphocytes infiltrates cancer glands in control group. (E) Few stromal cells with weak PGRMC1 expression (red arrows) in irradiated patients group. (F) Intensive expression of PGRMC1 by stroma (red arrows) in control group (magnification, ×200). FoxP3, forkhead box P3; Tregs, T regulatory cells; NK, natural killer; PGRMC1, progesterone receptor membrane component 1. 
Fig. 3.Box plot showing the number of FoxP3+ Tregs (A), CD56+ NK lymphocytes (C), and PGRMC1 (E) in Rad+ and control groups. The Kaplan-Mayer curves of FoxP3+ Tregs (B), CD56+ NK lymphocytes (D), and PGRMC1 (F) according to the threshold criteria. FoxP3, forkhead box P3; Tregs, T regulatory cells; NK, natural killer; PGRMC1, progesterone receptor membrane component 1. 
Fig. 4.Correlation plots of the irradiated group (A) and control group (B). CD56 indicates CD56+ natural killer lymphocytes; FoxP3, FoxP3+ Treg lymphocytes; PGRMC1, expression of progesterone receptor membrane component 1; FIGO stage, clinical stage by the International Federation of Gynecology and Obstetrics (2009); grade, grade of the endometrial carcinoma; unfavorable outcome, clinical unfavorable outcome (progression of endometrial carcinoma). 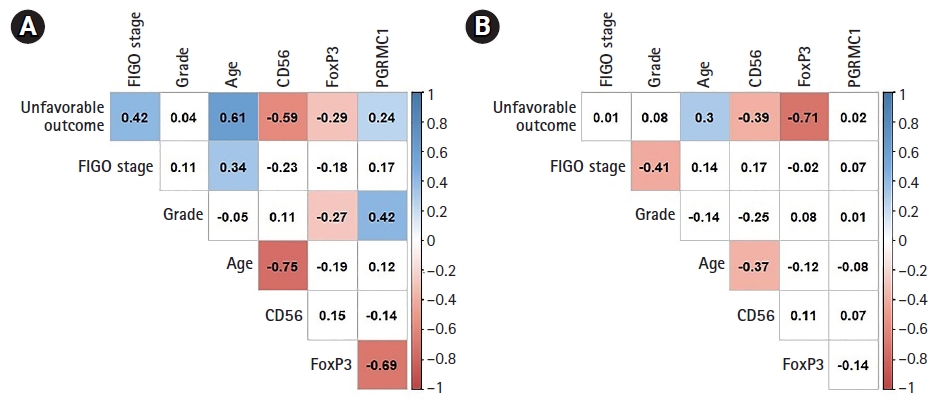
Table 1.Clinical characteristics of the patients
References1. Senol S, Sayar I, Ceyran AB, et al. Stromal clues in endometrial carcinoma: loss of expression of β-catenin, epithelial-mesenchymal transition regulators, and estrogen-progesterone receptor. Int J Gynecol Pathol 2016;35:238–48.
2. Kim JJ, Chapman-Davis E. Role of progesterone in endometrial cancer. Semin Reprod Med 2010;28:81–90.
3. Rodriguez AC, Blanchard Z, Maurer KA, Gertz J. Estrogen signaling in endometrial cancer: a key oncogenic pathway with several open questions. Horm Cancer 2019;10:51–63.
4. Romaniuk А, Lyndin M, Smiyanov V, et al. Primary multiple tumor with affection of the thyroid gland, uterus, urinary bladder, mammary gland and other organs. Pathol Res Pract 2017;213:574–9.
5. Roma-Rodrigues C, Raposo LR, Cabral R, Paradinha F, Baptista PV, Fernandes AR. Tumor microenvironment modulation via gold nanoparticles targeting malicious exosomes: implications for cancer diagnostics and therapy. Int J Mol Sci 2017;18:162.
6. Creutzberg CL, Nout RA. The role of radiotherapy in endometrial cancer: current evidence and trends. Curr Oncol Rep 2011;13:472–8.
7. Menon H, Ramapriyan R, Cushman TR, et al. Role of radiation therapy in modulation of the tumor stroma and microenvironment. Front Immunol 2019;10:193.
8. Wang HC, Chan LP, Cho SF. Targeting the immune microenvironment in the treatment of head and neck squamous cell carcinoma. Front Oncol 2019;9:1084.
9. Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science 2003;299:1057–61.
10. Shang B, Liu Y, Jiang SJ, Liu Y. Prognostic value of tumor-infiltrating FoxP3+ regulatory T cells in cancers: a systematic review and meta-analysis. Sci Rep 2015;5:15179.
11. de Jong RA, Leffers N, Boezen HM, et al. Presence of tumor-infiltrating lymphocytes is an independent prognostic factor in type I and II endometrial cancer. Gynecol Oncol 2009;114:105–10.
12. Levi I, Amsalem H, Nissan A, et al. Characterization of tumor infiltrating natural killer cell subset. Oncotarget 2015;6:13835–43.
13. Pietra G, Vitale C, Pende D, et al. Human natural killer cells: news in the therapy of solid tumors and high-risk leukemias. Cancer Immunol Immunother 2016;65:465–76.
14. Toffoli EC, Sheikhi A, Hoppner YD, et al. Natural killer cells and anti-cancer therapies: reciprocal effects on immune function and therapeutic response. Cancers (Basel) 2021;13:711.
15. Melaiu O, Lucarini V, Cifaldi L, Fruci D. Influence of the tumor microenvironment on NK cell function in solid tumors. Front Immunol 2020;10:3038.
16. Yang KL, Wang YS, Chang CC, et al. Reciprocal complementation of the tumoricidal effects of radiation and natural killer cells. PLoS One 2013;8:e61797.
17. Sorolla MA, Parisi E, Sorolla A. Determinants of sensitivity to radiotherapy in endometrial cancer. Cancers (Basel) 2020;12:1906.
18. Lee SR, Lee JG, Heo JH, et al. Loss of PGRMC1 delays the progression of hepatocellular carcinoma via suppression of pro-inflammatory immune responses. Cancers (Basel) 2021;13:2438.
19. Trabert B, Sherman ME, Kannan N, Stanczyk FZ. Progesterone and breast cancer. Endocr Rev 2020;41:320–44.
20. Kabe Y, Handa H, Suematsu M. Function and structural regulation of the carbon monoxide (CO)-responsive membrane protein PGRMC1. J Clin Biochem Nutr 2018;63:12–7.
21. Cahill MA, Jazayeri JA, Catalano SM, Toyokuni S, Kovacevic Z, Richardson DR. The emerging role of progesterone receptor membrane component 1 (PGRMC1) in cancer biology. Biochim Biophys Acta 2016;1866:339–49.
22. Hampton KK, Stewart R, Napier D, Claudio PP, Craven RJ. PGRMC1 elevation in multiple cancers and essential role in stem cell survival. Adv Lung Cancer (Irvine) 2015;4:37–51.
23. Lyzikova YA, Zinovkin DA, Pranjol MZ. Increase in FoxP3, CD56 immune cells and decrease in glands PGRMC1 expression in the endometrium are associated with recurrent miscarriages. Eur J Obstet Gynecol Reprod Biol 2020;245:121–6.
24. Koletsa T, Kotoula V, Koliou GA, et al. Prognostic impact of stromal and intratumoral CD3, CD8 and FOXP3 in adjuvantly treated breast cancer: do they add information over stromal tumor-infiltrating lymphocyte density? Cancer Immunol Immunother 2020;69:1549–64.
25. Lin ST, May EW, Chang JF, Hu RY, Wang LH, Chan HL. PGRMC1 contributes to doxorubicin-induced chemoresistance in MES-SA uterine sarcoma. Cell Mol Life Sci 2015;72:2395–409.
26. Arnold KM, Flynn NJ, Raben A, et al. The impact of radiation on the tumor microenvironment: effect of dose and fractionation schedules. Cancer Growth Metastasis 2018;11:1179064418761639.
27. Caetano MS, Younes AI, Barsoumian HB, et al. Triple therapy with MerTK and PD1 inhibition plus radiotherapy promotes abscopal antitumor immune responses. Clin Cancer Res 2019;25:7576–84.
28. Devaud C, Darcy PK, Kershaw MH. Foxp3 expression in T regulatory cells and other cell lineages. Cancer Immunol Immunother 2014;63:869–76.
29. Beauford SS, Kumari A, Garnett-Benson C. Ionizing radiation modulates the phenotype and function of human CD4+ induced regulatory T cells. BMC Immunol 2020;21:18.
30. Wirsdorfer F, Jendrossek V. The role of lymphocytes in radiotherapy-induced adverse late effects in the lung. Front Immunol 2016;7:591.
31. Liu S, Sun X, Luo J, et al. Effects of radiation on T regulatory cells in normal states and cancer: mechanisms and clinical implications. Am J Cancer Res 2015;5:3276–85.
32. Muroyama Y, Nirschl TR, Kochel CM, et al. Stereotactic radiotherapy increases functionally suppressive regulatory T cells in the tumor microenvironment. Cancer Immunol Res 2017;5:992–1004.
33. Venet F, Pachot A, Debard AL, et al. Human CD4+CD25+ regulatory T lymphocytes inhibit lipopolysaccharide-induced monocyte survival through a Fas/Fas ligand-dependent mechanism. J Immunol 2006;177:6540–7.
34. Lim SA, Kim TJ, Lee JE, et al. Ex vivo expansion of highly cytotoxic human NK cells by cocultivation with irradiated tumor cells for adoptive immunotherapy. Cancer Res 2013;73:2598–607.
35. Bigley AB, Simpson RJ. NK cells and exercise: implications for cancer immunotherapy and survivorship. Discov Med 2015;19:433–45.
36. Vivier E, Ugolini S. Natural killer cells: from basic research to treatments. Front Immunol 2011;2:18.
37. Diegeler S, Hellweg CE. Intercellular communication of tumor cells and immune cells after exposure to different ionizing radiation qualities. Front Immunol 2017;8:664.
38. Yoon SR, Kim TD, Choi I. Understanding of molecular mechanisms in natural killer cell therapy. Exp Mol Med 2015;47:e141.
39. Degos C, Heinemann M, Barrou J, et al. Endometrial tumor microenvironment alters human NK cell recruitment, and resident NK cell phenotype and function. Front Immunol 2019;10:877.
40. Kabe Y, Nakane T, Koike I, et al. Haem-dependent dimerization of PGRMC1/Sigma-2 receptor facilitates cancer proliferation and chemoresistance. Nat Commun 2016;7:11030.
41. Friel AM, Zhang L, Pru CA, et al. Progesterone receptor membrane component 1 deficiency attenuates growth while promoting chemosensitivity of human endometrial xenograft tumors. Cancer Lett 2015;356(2 Pt B):434–42.
42. He Y, Zhang P, Zhang D, et al. Combined assessment of low PGRMC1/positive ATP1A1 levels has enhanced prognostic value for renal cell carcinoma. Oncol Rep 2018;40:1467–76.
43. Ahmed IS, Rohe HJ, Twist KE, Craven RJ. PGRMC1 (progesterone receptor membrane component 1) associates with epidermal growth factor receptor and regulates erlotinib sensitivity. J Biol Chem 2010;285:24775–82.
44. Nijkamp MM, Span PN, Bussink J, Kaanders JH. Interaction of EGFR with the tumour microenvironment: implications for radiation treatment. Radiother Oncol 2013;108:17–23.
45. Zhang S, Liu W, Hu B, et al. Prognostic significance of tumor-infiltrating natural killer cells in solid tumors: a systematic review and meta-analysis. Front Immunol 2020;11:1242.
46. Tsai HW, Ho CL, Cheng SW, et al. Progesterone receptor membrane component 1 as a potential prognostic biomarker for hepatocellular carcinoma. World J Gastroenterol 2018;24:1152–66.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |