 |
 |
AbstractPurposeWe analyzed clinical results of CyberKnife (CK)-based stereotactic radiosurgery (SRS) or fractionated stereotactic radiotherapy (FSRT) in older patients (age Ōēź65 years) affected by brain metastases (BM) from non-small cell lung cancer (NSCLC).
Materials and MethodsForty-three older patients with 92 BM were treated with CK-based SRS/FSRT at our institution between 2009 and 2019. The end-point was overall survival (OS). Univariate and multivariate analyses were performed to identify the prognostic factors influencing OS. The in-field local control (IFLC) within the SRS/FSRT field was also assessed.
ResultsDuring a median follow-up period of 18 months, the median OS was 32 months. NSCLC-specific graded prognostic assessment (GPA) (p = 0.027) was an independent significant factor affecting OS in the multivariate analysis. The median IFLC period was 31 months, and the total BM volume (p = 0.025) appeared to be a significant feature of IFLC. No adverse events >grade 2 were reported after SRS/FSRT.
IntroductionBrain metastases (BMs) are the most common intracranial neoplasms and a significant cause of mortality in adults. In fact, up to 40% of patients affected by cancer will develop BM during their oncological history [1]. Recently, BM has frequently been diagnosed in long-term cancer survivors because of improved systemic therapies and early detection of BM by active surveillance using magnetic resonance imaging (MRI). The incidence of BM has shown a five-fold increase over the past few decades and is likely to continue to increase due to improvements in anti-cancer therapies [2]. It has been postulated that, in future, most new cancer diagnoses will be reported in the older population. Consequently, clinicians have been encountering a larger number of aging patients with BMs. Particularly, the incidence of BM derived from non-small cell lung cancer (NSCLC) is the highest, at approximately 20% [3].
The management of BM includes medical management, surgical resection, whole-brain radiotherapy (WBRT), stereotactic radiosurgery (SRS), and fractionated stereotactic radiotherapy (FSRT). When the number of BMs are limited, local treatments, such as surgical treatment, SRS, and FSRT can be used rather than WBRT. SRS and FSRT are effective treatments for patients with up to 10 BM, in terms of decreasing neurotoxicity, as the concept of oligometastases has emerged in BM [4-6].
Management of older patients with BMs remains a difficult issue, as it comprises an inhomogeneous population with diverse comorbidities and different physical statuses [7-9]. Age has been used as an important prognosticator in the recursive partitioning analysis (RPA) for classifying patients with BMs, as proposed by the Radiation Therapy Oncology Group, where patients Ōēź65 years old were classified as an intermediate risk group with RPA class II [7]. SRS and FSRT have increasingly been used for older patients with BMs, mainly because of the reduction in cognitive decline and the requirement of only a few treatment days [10-13].
To our knowledge, few studies have reported the efficacy of SRS in older patients with BMs using CyberKnife-based SRS or FSRT (CK-SRS/FSRT). Here, we analyzed the clinical results of CK-SRS/FSRT in an older cohort (Ōēź65 years old, RPA class II) affected by BMs arising from NSCLC.
Materials and Methods1. Study designThis retrospective study was approved by the Institutional Review Board of Inha University Hospital (No. 2022-09-006). Our institution administered CK-SRS/FSRT to patients with BM with a life expectancy of Ōēź3 months, Ōēż10 number of BM, and a diameter of BM Ōēż3 cm. CK-SRS/FSRT was administered on patients with BM not requiring decompressive surgery and those with Karnofsky performance status (KPS) scores Ōēź60. Those with leptomeningeal seeding were excluded.
For this study, we identified patients with BMs arising from NSCLC who were treated with upfront CK-SRS/FSRT between 2009 and 2019. After that, we extracted RPA class II patients who were aged Ōēź65 years and had a KPS Ōēź70 at the time of CK-SRS/FSRT. We scored patients with BM based on a diagnosis-specific graded prognostic assessment (GPA) index related to NSCLC [9].
2. CyberKnife treatmentAll patients received SRS/FSRT using a CyberKnife (Accuray Inc., Sunnyvale, CA, USA) equipped with a 6-MV linear accelerator mounted on a computer-controlled robotic arm with submillimeter accuracy. During CK-SRS/FSRT, each patient was placed in the supine position and fitted with a thermoplastic mask for immobilization. Computed tomography (CT) images of 1-mm slice thickness were fused with contrast-enhanced magnetic resonance images, Clinical target volume (CTV) was defined as the enhanced lesion observed on contrast-enhanced MRI. The planning target volume (PTV) was generated by adding a 2-mm margin to the CTV. The organs-at-risk (OARs), including the eyes, lenses, optic nerves, optic chiasm, brainstem, and spinal cord, were contoured. Plans were generated using a multiple inverse treatment-planning algorithm.
The total dose of CK-SRS/FSRT, with a range of 15ŌĆō32 Gy given in 1ŌĆō3 fractions, was prescribed to D90 (the radiation dose received by 90% of the PTV) based on BM size and proximity to OARs. To compensate for various dose-fractionation schedule, radiation doses were calculated as a biological effective dose (BED) based on the linear-quadratic equation with an alpha/beta ratio of 10. Target displacements caused by patient movement during treatment were automatically corrected. Stereoscopic X-ray images acquired during treatment were co-registered with a set of digitally reconstructed radiographs (DRRs) from dose-planning CT. The displacement vector was calculated by matching pairs of stereoscopic live images with the DRR.
3. OutcomesIn this study, the primary endpoint was overall survival (OS) following CK-SRS/FSRT, and the secondary endpoints were in-field local control (IFLC) within the CK-SRS/FSRT CTV and CK-SRS/FSRT-related toxicities. Additionally, we evaluated local tumor response within 6 months after CK-SRS/FSRT using the response assessment in neuro-oncology (RANO) criteria [14]. IFLC was defined as complete remission, partial remission, or stable disease, and progressive disease was categorized as in-field local failure.
4. Statistical analysisOS and IFLC were analyzed using the KaplanŌĆōMeier method. Univariate analysis was performed using log-rank tests to identify prognostic factors related to OS or IFLC. To assess the risk factors associated with OS or IFLC, multivariate Cox regression analyses were performed. IBM SPSS Statistics version 25.0 (IBM Corp., Armonk, NY, USA) was used, and a p <0.05 was defined as significant.
Results1. Patient and treatment characteristicsForty-three patients (92 BMs) were identified; their characteristics are listed in Table 1. The median patient age was 70 years (range, 65 to 89 years). Overall, 27 were males (63%). Of the 43 patients, 37 patients (86%) had Ōēż3 lesions. Thirty-two patients (74%) were diagnosed with adenocarcinoma. The median total BM volume per person was 1.70 cm3 (range, 0.07 to 81.84 cm3). With respect to diagnostic-specific GPA index scoring criteria based on lung cancer [9], three patients (7%) had a GPA of 0.5, 34 patients (79%) had a GPA of 1.0ŌĆō2.5, and six patients (14%) had a GPA of 3. Seventeen patients (40%) had extracranial disease present at the time of CK-SRS/FSRT.
Among the 92 BM lesions, 78 lesions (85%) were treated with CK-SRS and 14 (15%) were treated with CK-FSRT. The median prescribed dose was 22 Gy per fraction. The median BED was 81.6 Gy. The details of CK-SRS/FSRT for the 92 BM are summarized in Table 2.
2. Outcomes and factors affecting outcomesWithin a median follow-up time of 18 months (range, 2 to 104 months), OS at 12 and 24 months was 78.3% and 57.6%, respectively (Fig. 1A). The median OS period was 32 months (95% confidence interval [CI], 17ŌĆō46 months). In univariate analysis, GPA (p < 0.001, Fig 1B) and total metastatic tumor volume (p = 0.014, Fig 1C) were risk factors that significantly affected OS (Table 3). The number of BMs showed a borderline significance associated with OS (p = 0.095). Multivariate Cox regression analysis identified the GPA score as a powerful prognostic factor (p = 0.027) (Table 3).
Within 6 months after CK-SRS/FSRT, according to the RANO criteria for BM, complete remission was observed in 34.9% of patients (14/43), partial remission in 41.9% (18/43), and stable disease in 16.3% (7/43). Progressive disease was found in 3% of patients (3/43). IFLC within 6 months of CK-SRS/FSRT was observed in 40 of 43 patients (93.0%).
The overall IFLC at 12 months and 24 months was 84.5% and 58.4%, respectively (Fig. 2A). Additionally, the median IFLC period was 31 months (95% CI, 12ŌĆō50 months). Three clinical factors, including GPA (p < 0.001, Fig. 2B), total metastatic tumor volume (p < 0.001, Fig. 2C), and BED (p = 0.021, Fig. 2D) were found to be significant factors determining the IFLC period in the univariate analysis (Table 4). Subsequently, in the multivariate analysis, IFLC differed according to the total tumor volume (p = 0.025) (Table 4).
No acute or late adverse events higher than grade 2 were reported after CK-SRS/FSRT or during the follow-up.
Discussion and ConclusionFor cancer patients with a limited number of BMs, SRS/FSRT has increasingly been used in the initial management because this approach achieves excellent local control while avoiding the detrimental neurocognitive decline associated with WBRT. In particular, two randomized studies have shown no significant difference in OS or preservation of neurological function in patients with 1ŌĆō4 BMs who received WBRT with SRS, or SRS alone [5,6,15]. Since then, in keeping with the increase in the number of older cancer patients, several reports have explored the benefits in terms of OS in cohorts focusing on geriatric patients with BMs, although most physicians have historically considered cancer patients aged >65 years with BM as unfavorable candidates for active treatment, including surgery or SRS/FSRT. These studies have reported a median OS time of 7ŌĆō15 months and a 1-year IFLC rate of 80%ŌĆō99%, proving that SRS administration is a reasonable treatment for older patients with BM [10-13,16-19] (Table 5).
In line with these studies, our retrospective study showed the efficacy of upfront CK-SRS/FSRT in older patients with BM derived from NSCLC. In the present cohort, the OS and local control rate at 12 months after CK-SRS/FSRT were 78.3% and 84.5%, respectively. In addition, all patients in our study completed the planned CK-SRS/FSRT regimen without moderate or severe acute toxicity. NSCLC-specific GPA was demonstrated to be a strong prognosticator of OS, and the total BM volume was found to be a powerful factor in IFLC after adjusting for other confounding factors. In a previous study, patients aged >65 years with RPA class II had a median OS of 5 months [7]. Additionally, the GPA index showed an OS of 10 months in older patients affected by NSCLC [8,9]. When compared to these results, our study found a median OS of 32 months in Ōēź65-year-old patients with BM from NSCLC. This difference in survival time may be due to the good performance status (KPS Ōēź70), Ōēż10 BMs, with diameters Ōēż3 cm, and a short follow-up period.
Age, which is a common component of RPA and GPA [7-9], has been defined as an important prognostic factor in most patients with metastatic brain tumors. In many studies in which the proportion of older patients was low, outcomes were worse in older than in younger patients. However, some trials suggested that age should not be a criterion for excluding SRS in patients with BM [16,19]. Noel et al. [16] analyzed the outcomes of LINAC-based SRS for BMs in patients aged Ōēź65 years, by showing that the median OS was 8 months, which is comparable to that in the younger population. In addition, Higuchi et al. [19], in the prospective study (JLGK0901-Elderly), reported that the median survival time following Gamma Knife SRS was 10 months in older patients (aged Ōēź65 years), although the OS time was shorter than that in those younger than 65 years. The authors concluded that SRS is a favorable treatment option for older patients with BMs.
Korea's population is aging at an unprecedented rate [20]. The proportion of the population aged Ōēź65 years increased from 7% in 1999 to 11.8% in 2012 and is expected to increase to 20.8% by 2026, becoming a super-aged society. Population aging is a medical crisis related to high medical costs for individuals and long-term care costs for families and society [21]. Therefore, this study is valuable in this context, given that it is necessary to continue to consider optimal management for older patients, including those with BMs, to reduce the degree of medical crisis.
We found that the IFLC rate at 12 months was 84.5%, with a median IFLC period of 31 months. In the multivariate analysis related to IFLC, the total metastatic tumor volume was defined as a prognostic factor significantly associated with IFLC. Several studies have explored the efficacy of SRS as primary treatment for BMs, reporting a 12-month IFLC rate of 80%ŌĆō90%, with low IFLC observed in patients with larger lesions or volumes [17,22,23]. Vogelbaum et al. [22] reported a 12-month IFLC rate of 45% for lesions >2 cm as compared with 85% for lesions <2 cm. Chang et al. [23] reported a 12-month IFLC rate of 86% in tumors Ōēż1 cm in size and 56% in tumors >1 cm in BM treated with single-fraction SRS. In addition, Yomo et al. [17] defined a cumulative tumor volume (>2 mL) as the only predictor of a higher IFLC rate. Similar results were observed in our study, showing 73% and 53% of 12-month IFLC for cases with total BM volume of Ōēż3 mL versus >3 mL, respectively.
All patients completed the planned CK-SRS/FSRT without moderate or severe acute toxicity. Regarding CK-based therapy [24], CK is known as a dedicated SRS/FSRT device consisting of a compact and lightweight linear accelerator mounted on a robotic arm capable of movement with 6 degrees of freedom, allowing submillimeter targeting and unobstructed access to the entire body. Moreover, the device uses an image-guided control loop with target tracking that can be adjusted according to the patient's movement. According to these characteristics, in our study, CK-SRS/FSRT allowed the maintenance of the present quality of life by preventing neurological symptom deterioration or neurological catastrophe.
In recent decades, the circumstances surrounding cancers, including BM, have changed. As systemic therapies have become more efficacious in metastatic disease, the patterns of disease progression have changed with improvements in OS. Among them, oligometastatic BM is an emerging phenomenon, with limited or multiple BMs occurring while extra-cranial disease often remains under control. Yamamoto et al. [25] demonstrated that SRS might be a suitable approach for patients with up to 10 BMs, considering that it has fewer side effects than WBRT, by showing that the median OS in patients with 5ŌĆō10 BMs was 10.8 months, similar to that of a cohort with 2ŌĆō4 BMs. Yamamoto et al. [26] also performed a case-matched analysis comparing patients with 2ŌĆō9 BMs and Ōēź10 BMs, who were treated with SRS. The median OS or SRS-related complications did not differ between the cohorts. The authors suggested that even patients with Ōēź10 BMs could be suitable for SRS. In the new era with changing natural cancer history, establishing the proper approach for intracranial metastatic disease remains crucial [27-29]. Improving the preservation of a patientŌĆÖs quality of life and improving or maintaining tumor control are central dogmas in oncology research. Our study of CK-SRS/FSRT for BM with a focus on older patients represents an effort to achieve this trend and goal by de-escalating treatment volumes [29,30].
This study had several limitations. A significant issue is the patient selection bias inherent to retrospective studies. Our current study included older patients with a limited number of patients with BM who were able to tolerate the treatment. Given the retrospective nature of this study, we could not compare the potential role of SRS/FSRT with that of WBRT. Additionally, although the patient cohort had low heterogeneity overall as their BMs all arose from NSCLC, the relatively small number of patients and short-term follow-up period may have limited the statistical power of the analyses, leading to incomplete conclusions.
In conclusion, we investigated the efficacy of CK-SRS/FSRT for BMs in a cohort of older patients (Ōēź65 years of age) and suggested that CK-SRS/FSRT may be an effective treatment individuals of advanced age who have BMs. In particular, among these geriatric patients, those with a high GPA and low total BM tumor volume were considered favorable candidates for active BM treatment, such as SRS/FSRT, as these treatments contribute to longer survival and high local control, without severe side effects.
NotesStatement of Ethics This retrospective study was reviewed and approved by the Institutional Review Board of Inha University Hospital (No. 2022-09-006). Author Contributions Conceptualization, Lee J. Investigation and methodology, Lee J. Resources, Lee J, Kim HJ, Kim WC. Supervision, Kim HJ, Kim WC. Writing of the original draft, Lee J. Writing of the review and editing, Lee J, Kim HJ, Kim WC. Formal analysis, Lee J. Data curation, Lee J. All the authors have proofread the final version. Fig.┬Ā1.Overall survival following CK-based SRS/FSRT in the entire cohort (A), overall survival stratified according to non-small cell lung cancer-specific GPA (B), and total volume of brain metastases (C). CK, CyberKnife; SRS, stereotactic radiosurgery; FSRT, fractionated stereotactic radiotherapy; GPA, graded prognostic assessment. 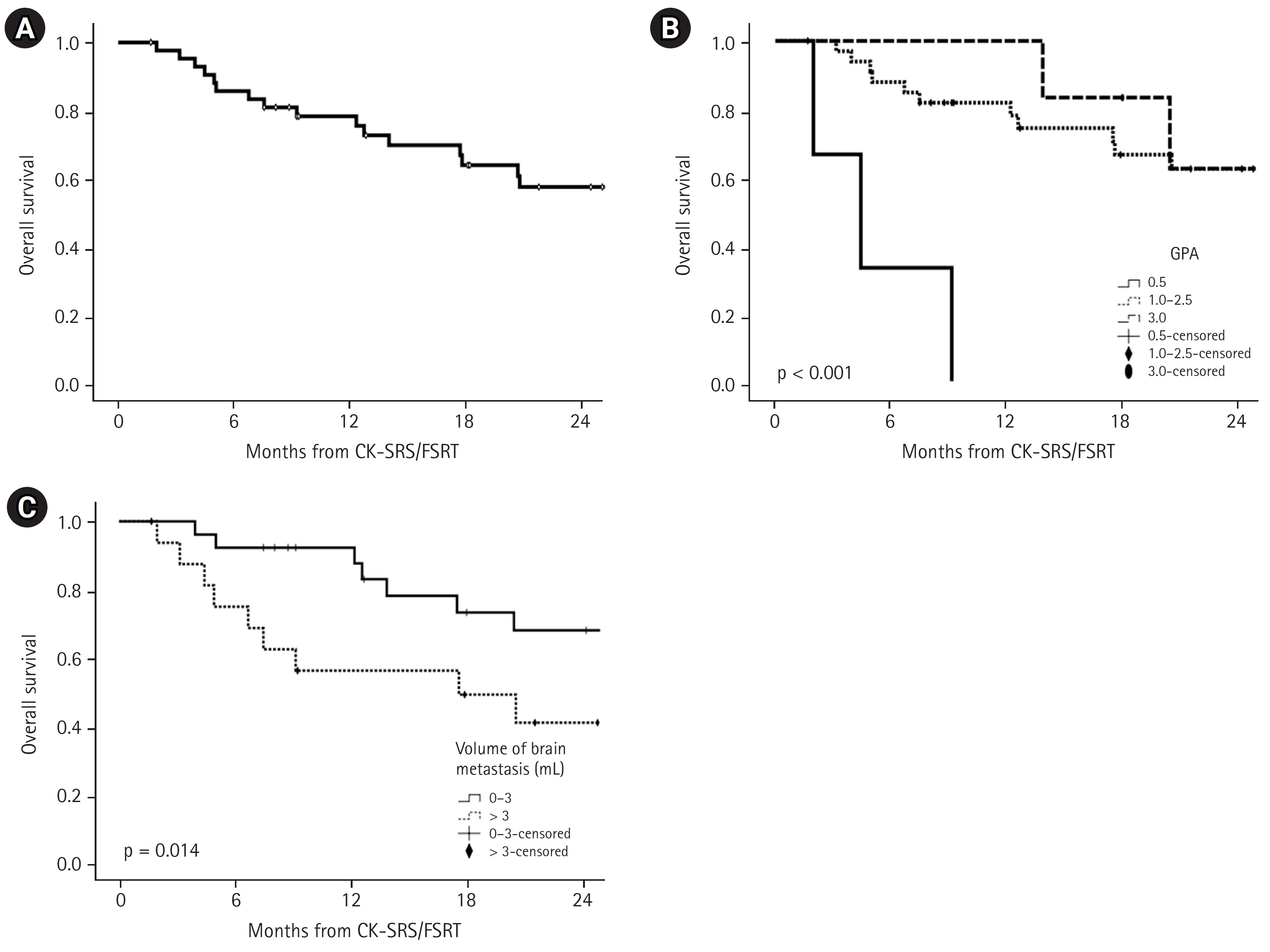
Fig.┬Ā2.In-field local control of brain metastasis treated with CK-based SRS/FSRT in the entire cohort (A), in-field local control stratified according to non-small cell lung cancer-specific GPA (B), total volume of brain metastases (C), and BED (D). CK, CyberKnife; SRS, stereotactic radiosurgery; FSRT, fractionated stereotactic radiotherapy; GPA, graded prognostic assessment; BED, biological effective dose based on the linear-quadratic equation with an alpha/beta ratio of 10. 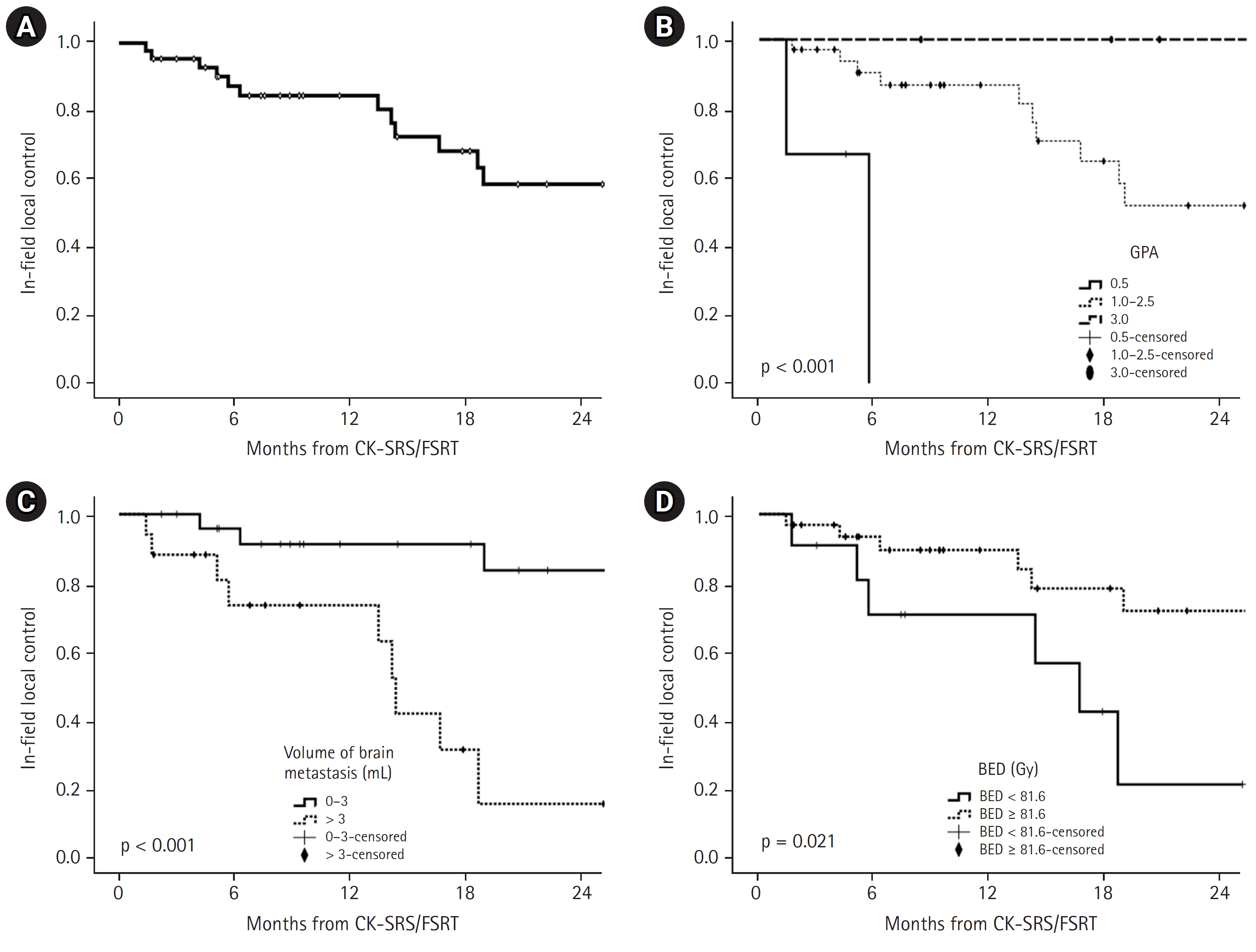
Table┬Ā1.Patient characteristics (n = 43) Table┬Ā2.Characteristics of CyberKnife-based SRS/FSRT (n = 92) Table┬Ā3.Prognostic factors related to overall survival Table┬Ā4.Prognostic factors related to in-field local control Table┬Ā5.Publications on SRS or FSRT for elderly patients with brain metastases
References2. Yancik R, Ries LA. Cancer in older persons: an international issue in an aging world. Semin Oncol 2004;31:128ŌĆō36.
3. Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol 2004;22:2865ŌĆō72.
4. Scoccianti S, Ricardi U. Treatment of brain metastases: review of phase III randomized controlled trials. Radiother Oncol 2012;102:168ŌĆō79.
5. Brown PD, Jaeckle K, Ballman KV, Farace E, Cerhan JH, Anderson SK, et al. Effect of radiosurgery alone vs radiosurgery with whole brain radiation therapy on cognitive function in patients with 1 to 3 brain metastases: a randomized clinical trial. JAMA 2016;316:401ŌĆō9.
6. Aoyama H, Shirato H, Tago M, Nakagawa K, Toyoda T, Hatano K, et al. Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. JAMA 2006;295:2483ŌĆō91.
7. Gaspar L, Scott C, Rotman M, Asbell S, Phillips T, Wasserman T, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys 1997;37:745ŌĆō51.
8. Sperduto PW, Berkey B, Gaspar LE, Mehta M, Curran W. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG database. Int J Radiat Oncol Biol Phys 2008;70:510ŌĆō4.
9. Sperduto PW, Chao ST, Sneed PK, Luo X, Suh J, Roberge D, et al. Diagnosis-specific prognostic factors, indexes, and treatment outcomes for patients with newly diagnosed brain metastases: a multi-institutional analysis of 4,259 patients. Int J Radiat Oncol Biol Phys 2010;77:655ŌĆō61.
10. Minniti G, Esposito V, Clarke E, Scaringi C, Bozzao A, Lanzetta G, et al. Stereotactic radiosurgery in elderly patients with brain metastases. J Neurooncol 2013;111:319ŌĆō25.
11. Kim SH, Weil RJ, Chao ST, Toms SA, Angelov L, Vogelbaum MA, et al. Stereotactic radiosurgical treatment of brain metastases in older patients. Cancer 2008;113:834ŌĆō40.
12. Gregucci F, Fiorentino A, Corradini S, Figlia V, Mazzola R, Ricchetti F, et al. Linac-based radiosurgery or fractionated stereotactic radiotherapy with flattening filter-free volumetric modulated arc therapy in elderly patients: a mono-institutional experience on 110 brain metastases. Strahlenther Onkol 2019;195:218ŌĆō25.
13. Yamamoto M, Serizawa T, Sato Y, Higuchi Y, Kasuya H, Barfod BE. Stereotactic radiosurgery for brain metastases: a retrospective cohort study comparing treatment results between two lung cancer patient age groups, 75 years or older vs 65-74 years. Lung Cancer 2020;149:103ŌĆō12.
14. Lin NU, Lee EQ, Aoyama H, Barani IJ, Barboriak DP, Baumert BG, et al. Response assessment criteria for brain metastases: proposal from the RANO group. Lancet Oncol 2015;16:e270.
15. Kocher M, Soffietti R, Abacioglu U, Vill├Ā S, Fauchon F, Baumert BG, et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol 2011;29:134ŌĆō41.
16. Noel G, Bollet MA, Noel S, Feuvret L, Boisserie G, Tep B, et al. Linac stereotactic radiosurgery: an effective and safe treatment for elderly patients with brain metastases. Int J Radiat Oncol Biol Phys 2005;63:1555ŌĆō61.
17. Yomo S, Hayashi M. Is upfront stereotactic radiosurgery a rational treatment option for very elderly patients with brain metastases?: a retrospective analysis of 106 consecutive patients age 80 years and older. BMC Cancer 2016;16:948.
18. Chen L, Shen C, Redmond KJ, Page BR, Kummerlowe M, Mcnutt T, et al. Use of stereotactic radiosurgery in elderly and very elderly patients with brain metastases to limit toxicity associated with whole brain radiation therapy. Int J Radiat Oncol Biol Phys 2017;98:939ŌĆō47.
19. Higuchi Y, Yamamoto M, Serizawa T, Sato Y, Shuto T, Akabane A, et al. Stereotactic radiosurgery in elderly patients with brain metastases: comparison with non-elderly patients using database of a multi-institutional prospective observational study (JLGK0901-Elderly). J Neurooncol 2019;144:393ŌĆō402.
20. Kim HJ, Min KB, Min JY. Healthy aging predicts mortality risks: results from the Korean longitudinal study of aging (2006-2014). Arch Gerontol Geriatr 2021;94:104333.
21. Hyun KR, Kang S, Lee S. Population aging and healthcare expenditure in Korea. Health Econ 2016;25:1239ŌĆō51.
22. Vogelbaum MA, Angelov L, Lee SY, Li L, Barnett GH, Suh JH. Local control of brain metastases by stereotactic radiosurgery in relation to dose to the tumor margin. J Neurosurg 2006;104:907ŌĆō12.
23. Chang EL, Hassenbusch SJ 3rd, Shiu AS, Lang FF, Allen PK, Sawaya R, et al. The role of tumor size in the radiosurgical management of patients with ambiguous brain metastases. Neurosurgery 2003;53:272ŌĆō81.
24. Ding C, Saw CB, Timmerman RD. Cyberknife stereotactic radiosurgery and radiation therapy treatment planning system. Med Dosim 2018;43:129ŌĆō40.
25. Yamamoto M, Serizawa T, Shuto T, Akabane A, Higuchi Y, Kawagishi J, et al. Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901): a multi-institutional prospective observational study. Lancet Oncol 2014;15:387ŌĆō95.
26. Yamamoto M, Kawabe T, Sato Y, Higuchi Y, Nariai T, Watanabe S, et al. Stereotactic radiosurgery for patients with multiple brain metastases: a case-matched study comparing treatment results for patients with 2-9 versus 10 or more tumors. J Neurosurg 2014;121 Suppl:16ŌĆō25.
27. Fecci PE, Champion CD, Hoj J, McKernan CM, Goodwin CR, Kirkpatrick JP, et al. The evolving modern management of brain metastasis. Clin Cancer Res 2019;25:6570ŌĆō80.
28. Shen CJ, Lim M, Kleinberg LR. Controversies in the therapy of brain metastases: shifting paradigms in an era of effective systemic therapy and longer-term survivorship. Curr Treat Options Oncol 2016;17:46.
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |