 |
 |
AbstractPurposeSurface-guided radiation therapy is an image-guided method using optical surface imaging that has recently been adopted for patient setup and motion monitoring during treatment. We aimed to determine whether the surface guide setup is accurate and efficient compared to the skin-marking guide in prostate cancer treatment.
Materials and MethodsThe skin-marking setup was performed, and vertical, longitudinal, and lateral couch values (labeled as "M") were recorded. Subsequently, the surface-guided setup was conducted, and couch values (labeled as "S") were recorded. After performing cone-beam computed tomography (CBCT), the final couch values was recorded (labeled as "C"), and the shift value was calculated (labeled as "Gap (M-S)," "Gap (M-C)," "Gap (S-C)") and then compared. Additionally, the setup times for the skin marking and surface guides were also compared.
ResultsOne hundred and twenty-five patients were analyzed, totaling 2,735 treatment fractions. Gap (M-S) showed minimal differences in the vertical, longitudinal, and lateral averages (-0.03 cm, 0.07 cm, and 0.06 cm, respectively). Gap (M-C) and Gap (S-C) exhibited a mean difference of 0.04 cm (p = 0.03) in the vertical direction, a mean difference of 0.35 cm (p = 0.52) in the longitudinal direction, and a mean difference of 0.11 cm (p = 0.91) in the lateral direction. There was no correlation between shift values and patient characteristics. The average setup time of the skin-marking guide was 6.72 minutes, and 7.53 minutes for the surface guide.
ConclusionThere was no statistically significant difference between the surface and skin-marking guides regarding final CBCT shift values and no correlation between translational shift values and patient characteristics. We also observed minimal difference in setup time between the two methods. Therefore, the surface guide can be considered an accurate and time-efficient alternative to skin-marking guides.
IntroductionThe precision of radiation therapy administration has been enhanced by technological progress, and the growing adoption of hypofractionation has underscored the significance of ensuring accurate patient setup [1-3]. The most commonly used method for patient posture setup in clinics is the laser guide with skin marking, which involves drawing lines on the body of the patient or applying a tattooed reference point during the simulation and then aligning it with a laser in the treatment room. However, skin markings can be erased during conventional fraction treatment, causing inconvenience to patients who cannot wash their bodies. In case the skin markings are gone, the simulation and treatment plan must be re-evaluated, which can be time- and cost-inefficient. Additionally, tattoos are invasive and can cause physical or psychological discomfort if they are permanent [4]. Moreover, due to the elasticity of the skin, even when the laser is aligned with the skin marking, there remains a possibility that the setup may not be entirely accurate.
Surface-guided radiotherapy (SGRT) utilizes three light projectors and an optical camera to generate a 3D map of the body contour of the patient, which is compared to a reference surface image. The software analyzes the differences in real-time and displays them as translational and rotational delta values. Since the beam is non-ionizing, it allows for real-time adjustment of the positional setup during treatment, improving accuracy and efficiency [5]. In addition, studies on the application of the surface guide to patient identification, patient safety [6], and motion management are ongoing [2,7], and gating radiotherapy as well as set up in head and neck cancer [8-10], stereotactic radiosurgery, and breast cancer [7,11-15]. This study aims to measure and compare the accuracy of the surface-guided setup. We aim to confirm that surface guide setup is a comfortable and accurate method for patients, which does not require additional effort.
Materials and MethodsWe analyzed 2,735 external radiation treatments administered to 125 patients who underwent external beam radiation therapy (EBRT) for prostate cancer at Asan Medical Center between August 2021 and February 2022. Patient data for this study were collected through an Institutional Review Board (IRB)-approved retrospective chart review (IRB No. S2023-0461-0001). After the range of treatment was confirmed by the physician, the patients lay down on their backs with pillows under their heads and ankles and arms raised above their chests. The balloon catheter was then inserted into the rectum, adjusted to fit the patient, inflated with 80 mL of air, and secured. The scanning range was set to the proximal femur from T11. Administration of contrast required the scan to use a 2.5-mm slice after a delay of approximately 180–210 seconds. Skin marking was performed after computed tomography (CT) simulation (Discovery RT; GE HealthCare, Waukesha, WI, USA) for all treated patients, with a horizontal line connecting both femur heads, mid-pelvis lines on both lateral sides, and a midline based on the patient's pubic symphysis. Skin markings were applied to four locations, and a CT simulation was performed. All patients were treated with Halcyon equipped with the AlignRT (Vision RT Inc., London, UK) surface guide system.
The patient was initially positioned and aligned during each treatment using the skin marking method drawn during CT simulation. After turning off the laser, the patient was gently aided to stand up, and then the surface guide was setup using the AlignRT system. Then, based on the surface guide couch value, cone-beam computed tomography (CBCT) image guide was performed. The couch values are labeled as "M" during the skin marking setup, "S" during the SGRT setup, and "C" after CBCT and couch shift. Gap (M-C) represents the difference between "M" and "C," Gap (S-C) represents the difference between "S" and "C," and Gap (M-S) represents the difference between "M" and "S" (Fig. 1). AlignRT comprises three light projectors and one optical camera installed on the ceiling of the treatment room. The optical camera analyzes the light beams emitted from the projectors to create a 3D map of the body contour, then compares it with the reference image's body contour (Fig. 2A). A reference surface image was created from the external surface of the CT simulation scan and imported through the DICOM file. The AlignRT software compares real-time surfaces and displays any differences as three translational (ventral, lateral, longitudinal) and three rotational (yaw, roll, pitch) delta values. During daily treatment, the translational delta values were kept within 3 mm, and the rotational delta values were setup within 1°. The translational shift was accomplished via couch movement for patient setup, while the rotational shift was performed manually (Fig. 2B). Both lateral thighs were set as region-of-interest (Fig. 2C). The setup time was measured from the start of surface guide setup to the final shift using CBCT, just before beam irradiation, and was compared with the time taken in other treatment rooms where prostate cancer was treated with TomoTherapy (Accuray Inc., Sunnyvale, CA, USA).
Statistical analysis was performed using R statistics version 4.1.3 and Microsoft Excel 2020. The vertical, longitudinal, and lateral couch values of the skin-marking and surface-guide setups were analyzed based on surface-guide couch values. The couch values of the skin-marking setup and the surface-guide setup were indirectly compared by analyzing the couch value obtained after performing CBCT with image guidance. A t-test was used to compare the difference in the final shift value between the skin-marking and surface-guide methods and the setup time for each method.
In this study, we examine the translation vector by calculating its magnitude using the square root of the sum of the squares in each direction. This can be expressed mathematically as follows:
ResultsThe median age of the patients was 71.3 years (range, 45 to 88 years), with a median height of 165.5 cm (range, 152.1 to 182.0 cm) and a median body mass index (BMI) of 25.5 kg/m2 (range, 19.5 to 35.5 kg/m2) (Table 1).
Gap (M-S) showed that the vertical, long, and lateral averages differed by only -0.03 cm, 0.07 cm, and 0.06 cm, respectively (Fig. 3A). Furthermore, the standard deviation was within 0.5, confirming the consistency of the results. In the vertical direction, the mean difference between Gap (M-C) (range, -0.05 to 0.65) and Gap (S-C) (range, -0.05 to 0.76) was 0.04 cm (p = 0.03), and in the longitudinal direction, the mean difference between Gap (M-C) (range, -0.31 to 0.47) and Gap (S-C) (range, -0.29 to 0.88) was 0.35 cm (p = 0.52), and the lateral direction showed a mean difference of 0.11 cm (range, -0.24 to 0.72 and -0.24 to 0.75, respectively) (p = 0.91) (Fig. 3B, 3C).
The mean translation vector value of Gap (M-C) was 0.39 cm (range, 0.08 to 0.78), while the mean translation vector value of Gap (S-C) was 0.43 cm (range, 0.08 to 0.89). The mean difference between the two groups was 0.04 cm (p < 0.0001; 95% confidence interval, 0.028–0.056). Multiple linear regression analysis was conducted, revealing no significant associations between the three-directional translation vector of Gap (M-C) and patient age (p = 0.931), height (p = 0.516), and BMI (p = 0.107), as well as no significant associations between the three-directional translation vector of Gap (S-C) and patient age (p = 0.623), height (p = 0.632), and BMI (p = 0.179).
When comparing Gap (S-C) from the start to the end of the treatment, no significant differences were observed in the vertical (p = 0.932), longitudinal (p = 0.562), and lateral (p = 0.85) directions. Additionally, an analysis of the association with weight change yielded no statistically significant findings in the vertical (p = 0.378), longitudinal (p = 0.264), and lateral (p = 0.455) directions.
The setup time for Halcyon with the surface guide was 7.53 minutes (range, 5 to 10 minutes) and 6.73 minutes (range, 5 to 8 minutes) for TomoTherapy with skin marking, resulting in a difference of 0.8 minutes between the two groups (p < 0.0001) (Fig. 3D).
Discussion and ConclusionComparison of the skin-marking and surface-guide setups after CBCT showed a negligible difference in the couch shift value of less than 1 mm in all directions, unaffected by patient age, BMI, and height.
A previous study on surface-guide setup in radiotherapy for prostate cancer reported a median vector offset of 0.47 cm, comparable to our finding of 0.43 cm. The study also noted a faster setup time than with the skin-marking guide [17]. Additionally, several studies have shown that applying surface guides to the pelvis and lower extremities results in a translational difference of 0.6 cm [18], whereas in the head and neck it ranges from 0.1 to 0.3 cm [5,9], and 0.24 to 0.87 cm in the breast and thorax [14,15,18,19]. A difference as large as 1 cm can be observed in the abdomen [5,18] (Table 2). In contrast to the breast, head, and neck, surface contour is not a reliable indicator of the position of internal organs in the prostate.
To evaluate accuracy of surface guides in prostate setup, we conducted a study that revealed a disparity between the surface-guide setup and the final shift value that was similar to that observed in other body areas. Our findings confirm that surface guides are a reasonably accurate method for setup in prostate cancer treatment compared with skin marking. However, several studies have reported that the translational difference of skin marking is greater than that of surface guide [14,15,17-19], suggesting that the latter is a more accurate method. Our study observed a translational difference of 0.39 cm in the skin-marking setup compared with the 0.52 cm reported previously [17]. This suggests that our setup method, based on drawing a long reference line on the patient's body, is a more accurate alternative to using a tattoo point. However, if the reference skin marking is long, the setup is accurate, but the patient's discomfort may increase. Therefore, this study confirms that SGRT is accurate enough to replace our accurate long reference line, which may reduce patient discomfort.
In addition, our study confirms that the differences in the vertical direction were greater compared to CBCT, indicating similar findings for both skin marking and SGRT. These observations may be attributed to couch sagging in Halcyon, highlighting the potential for enhanced accuracy with the availability of in-bore type SGRT.
While this study focused on applying surface guides as an alternative to skin marking in prostate cancer treatment, surface guides have potential uses in various other applications. For example, these systems offer valuable applications, such as maskless setup for head and neck treatments and reliable patient identification [8-10], wherein the body contour of the patient can serve as a means of identification [1-3,12]. Additionally, when combined with four-dimensional computed tomography (4DCT) imaging, surface guidance enhances treatment accuracy through the gating system, ensuring precise targeting [2,8,20]. This approach also enables us to capture vital patient information, including identification and weight loss, while adapting radiotherapy to account for the daily size changes of surface-adjacent tumors. The integration of augmented reality holds promises in projecting internal patient data onto the surface, further augmenting the setup process [1,3,12].
There are several limitations to the present study. First, performing two setups and to CBCTs would ensure accurate comparison of the two methods; however, given the increased exposure to radiation and potential inconvenience to the patient, we adopted a different approach. To mitigate bias introduced by the initial setup, the patient was stood up and underwent a reset procedure with SGRT for the second setup after completing the first setup with skin marking. Second, performing two setups on the same patient while measuring the setup time would improve the accuracy of our data. However, setup times are typically very short (usually 20 seconds to 1 minute), so accurately measuring them can be challenging. To address this, we determined the overall treatment preparation time by measuring the time from setup initiation to completion just after CBCT image guidance. We then compared this time to that of a prostate treatment patient receiving TomoTherapy with a skin-marking guide. To ensure a more accurate comparison, it is essential to use the same equipment. Nevertheless, our institution had two dedicated treatment rooms, SGRT-Halcyon and skin marking with TomoTherapy, specifically for prostate cancer patients during the study, and thus this comparison was conducted based on these methods. The difference between the surface guide and skin marking was less than 1 minute. Notably, these data were collected when the device was first introduced, so some proficiency issues may have occurred. However, we anticipate that there is currently very little disparity in setup times between the two methods.
In conclusion, surface-guided radiotherapy in prostate EBRT is reliable and could replace skin marking as an accurate and efficient patient identification and setup method.
NotesStatement of Ethics This study was approved by the Institutional Review Board at Asan Medical Center (IRB No. S2023-0461-0001). Fig. 1.Treatment sequence: the skin-marking guide is followed by the surface guide, then the image guide is performed by taking a cone-beam computed tomography. 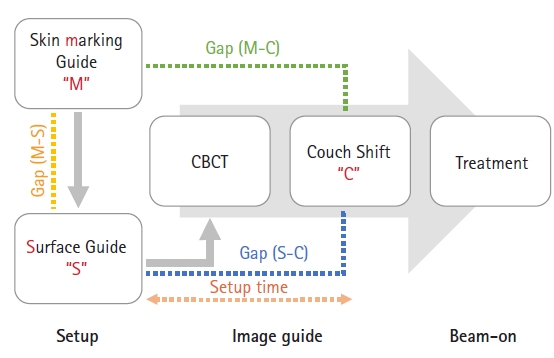
Fig. 2.The beams from the three beam projectors installed on the ceiling (A) are analyzed by an optical camera and compared with the reference image obtained from the CT simulation (B) to perform the setup. The region-of-interest is both lateral thighs (C). 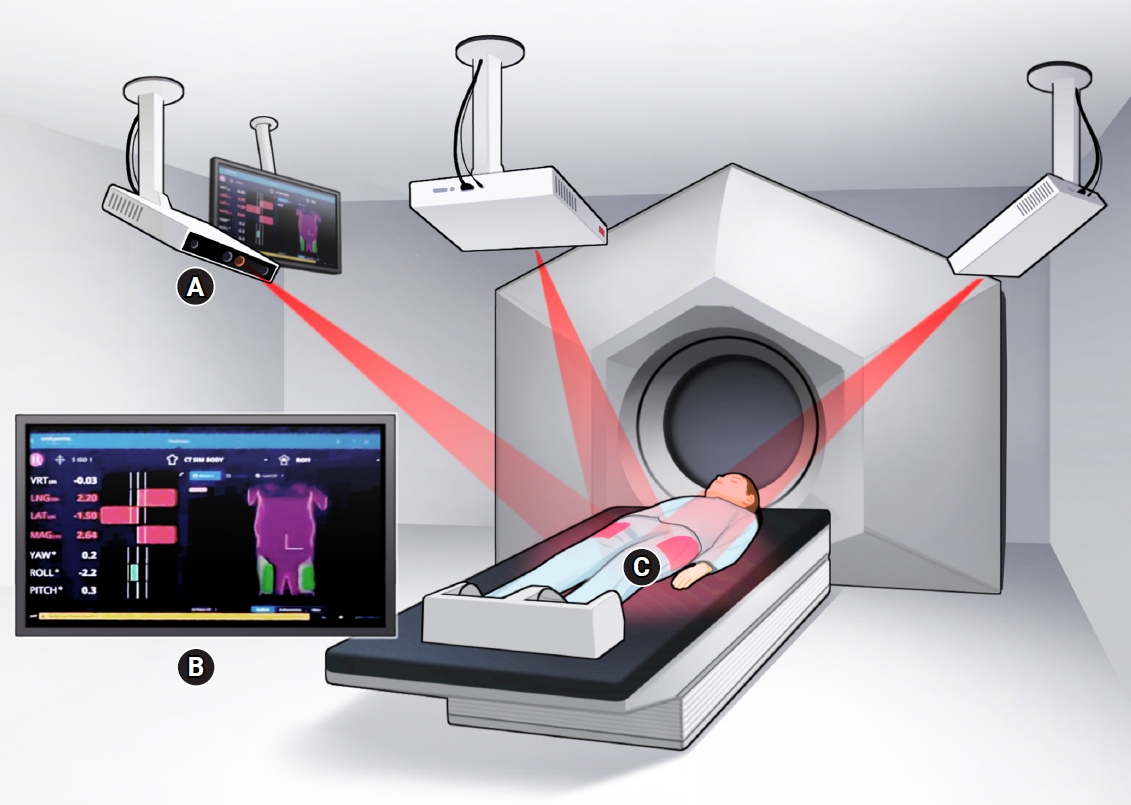
Fig. 3.The following four aspects were evaluated: (A) three-directional differences between the skin marking and surface guide, (B) differences between the skin marking and CBCT shift values, (C) differences between the surface guide and CBCT shift values, and (D) setup time of the surface-guide technique using Halcyon compared with the skin-marking guide using TomoTherapy. CBCT, cone-beam computed tomography; SGRT, surface-guided radiation therapy. 
Table 1.Patient characteristics (n = 125) Table 2.Translational differences of skin marking and surface guide
References1. Batista V, Meyer J, Kugele M, Al-Hallaq H. Clinical paradigms and challenges in surface guided radiation therapy: where do we go from here? Radiother Oncol 2020;153:34–42.
2. Paolani G, Strolin S, Santoro M, et al. A novel tool for assessing the correlation of internal/external markers during SGRT guided stereotactic ablative radiotherapy treatments. Phys Med 2021;92:40–51.
3. Al-Hallaq H, Batista V, Kugele M, Ford E, Viscariello N, Meyer J. The role of surface-guided radiation therapy for improving patient safety. Radiother Oncol 2021;163:229–36.
4. Rigley J, Robertson P, Scattergood L. Radiotherapy without tattoos: could this work? Radiography (Lond) 2020;26:288–93.
5. Haraldsson A, Ceberg S, Ceberg C, Back S, Engelholm S, Engstrom PE. Surface-guided tomotherapy improves positioning and reduces treatment time: a retrospective analysis of 16 835 treatment fractions. J Appl Clin Med Phys 2020;21:139–48.
6. Wiant DB, Verchick Q, Gates P, et al. A novel method for radiotherapy patient identification using surface imaging. J Appl Clin Med Phys 2016;17:271–8.
7. Song Y, Zhai X, Liang Y, Zeng C, Mueller B, Li G. Evidence-based region of interest (ROI) definition for surface-guided radiotherapy (SGRT) of abdominal cancers using deep-inspiration breath-hold (DIBH). J Appl Clin Med Phys 2022;23:e13748.
8. Zhao B, Park YK, Gu X, Reynolds R, Timmerman R, Sher DJ. Surface guided motion management in glottic larynx stereotactic body radiation therapy. Radiother Oncol 2020;153:236–42.
9. Lee SK, Huang S, Zhang L, et al. Accuracy of surface-guided patient setup for conventional radiotherapy of brain and nasopharynx cancer. J Appl Clin Med Phys 2021;22:48–57.
10. Zhao B, Maquilan G, Jiang S, Schwartz DL. Minimal mask immobilization with optical surface guidance for head and neck radiotherapy. J Appl Clin Med Phys 2018;19:17–24.
11. Hoisak JD, Pawlicki T. The role of optical surface imaging systems in radiation therapy. Semin Radiat Oncol 2018;28:185–93.
12. Freislederer P, Kugele M, Ollers M, et al. Recent advanced in surface guided radiation therapy. Radiat Oncol 2020;15:187.
13. Svestad JG, Heydari M, Mikalsen SG, Flote VG, Nordby F, Hellebust TP. Surface-guided positioning eliminates the need for skin markers in radiotherapy of right sided breast cancer: a single center randomized crossover trial. Radiother Oncol 2022;177:46–52.
14. Kugele M, Mannerberg A, Norring Bekke S, et al. Surface guided radiotherapy (SGRT) improves breast cancer patient setup accuracy. J Appl Clin Med Phys 2019;20:61–8.
15. Jimenez RB, Batin E, Giantsoudi D, et al. Tattoo free setup for partial breast irradiation: a feasibility study. J Appl Clin Med Phys 2019;20:45–50.
16. Naidoo W, Leech M. Feasibility of surface guided radiotherapy for patient positioning in breast radiotherapy versus conventional tattoo-based setups: a systematic review. Tech Innov Patient Support Radiat Oncol 2022;22:39–49.
17. Mannerberg A, Kugele M, Hamid S, et al. Faster and more accurate patient positioning with surface guided radiotherapy for ultra-hypofractionated prostate cancer patients. Tech Innov Patient Support Radiat Oncol 2021;19:41–5.
18. Stanley DN, McConnell KA, Kirby N, Gutierrez AN, Papanikolaou N, Rasmussen K. Comparison of initial patient setup accuracy between surface imaging and three point localization: a retrospective analysis. J Appl Clin Med Phys 2017;18:58–61.
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |