 |
 |
AbstractPurposeTo report the results of stereotactic body radiotherapy (SBRT) for unresectable primary or recurrent cholangiocarcinoma.
Materials and MethodsFrom January 2005 through August 2013, 58 patients with unresectable primary (n = 28) or recurrent (n = 30) cholangiocarcinoma treated by SBRT were retrospectively analyzed. The median prescribed dose was 45 Gy in 3 fractions (range, 15 to 60 Gy in 1-5 fractions). Patients were treated by SBRT only (n = 53) or EBRT + SBRT boost (n = 5). The median tumor volume was 40 mL (range, 5 to 1,287 mL).
ResultsThe median follow-up duration was 10 months (range, 1 to 97 months). The 1-year, 2-year overall survival rates, and median survival were 45%, 20%, and 10 months, respectively. The median survival for primary group and recurrent group were 5 and 13 months, respectively. Local control rate at 1-year and 2-year were 85% and 72%, respectively. Disease progression-free survival rates at 1-year and 2-year were 26% and 23%, respectively. In univariate analysis, ECOG performance score (0-1 vs. 2-3), treatment volume (<50 vs. ≥50 mL), and pre-SBRT CEA level (<5 vs. ≥5 ng/mL) were significant in overall survival rate. In multivariate analysis, ECOG score (p = 0.037) and tumor volume (p = 0.030) were statistically significant. In the recurrent tumor group, patients with >12 months interval from surgery to recurrence showed statistically significant higher overall survival rate than those with ≤12 months (p = 0.026). Six patients (10%) experienced ≥grade 3 complications.
IntroductionCholangiocarcinoma is a rare malignancy originating from the epithelial cells of the bile ducts. It is classically classified as intrahepatic and extrahepatic tumor according to their anatomical origins. Currently, surgical resection provides the only possibility of cure. However, the majority of patients present with unresectable disease and therefore have dismal prognosis. Resectability rates are generally low, usually less than 30% [1]. Unresectable cholangiocarcinoma has a dismal prognosis with a median survival of only 3-6 months [2]. For patients with cholangiocarcinoma who are unsuitable for surgery, other local alternative treatment options include external beam radiotherapy (EBRT), radiofrequency ablation or transarterial chemoembolization [3]. Yet even with this diverse list of treatment modalities, the median survival for locally-advanced or recurrent cholangiocarcinoma is approximately 9 months, and 5-year survival is less than 5% [3]. Early results using EBRT and brachytherapy have demonstrated a dose-response relationship [4,5,6]. This has allowed a potential role of dose-escalation as a means of improving disease control and survival.
Stereotactic body radiotherapy (SBRT) can be a promising treatment modality, which involves the delivery of very high doses with high precision. In this study, we present the clinical outcomes and toxicity associated with SBRT for unresectable or recurrent cholangiocarcinoma.
Materials and Methods1. PatientsWe retrospectively reviewed the records of 58 patients with unresectable primary (n = 28) or recurrent (n = 30) cholangiocarcinoma who were treated with SBRT at our institution between January 2005 and August 2013. Primary tumors were found in liver (n = 27) or liver + enlarged lymph node (n = 1). Recurrent tumors were found in liver (n = 12), para-aortic lymph node (n = 17), or liver + enlarged lymph node (n = 1). All patients presented the Eastern Cooperative Oncology Group (ECOG) performance status of 0-3 and assessed by computed tomography (CT), magnetic resonance imaging, or positron emission tomography/computed tomography before SBRT. The decision 'inoperable tumor' and the following decision for SBRT was made in an interdisciplinary tumor board. The diagnosis was confirmed by histology or image study. The demographic data as well as the tumor and therapy data of the patients are depicted in Table 1. Fifty-three patients were treated with SBRT alone and 5 patients with EBRT + SBRT boost. Of the 30 patients with recurrent tumor, 9 patients previously received RT (range, 40-63 Gy/20-35 fractions) to the primary cancer lesion (liver). Among them, there was an overlap in the previous and current irradiated lesions in 6 patients.
2. The EBRT techniqueFive patients received EBRT followed by SBRT boost. The patients were selected by radiation oncologist's discretion. Total 38-44 Gy (median, 40 Gy) of EBRT was delivered with conventional fractionation (2 Gy). EBRT was administered via 3-dimensional conformal RT. All patients underwent CT simulation in the supine position with arms elevated above the head. A CMS XiO treatment planning system (CMS Inc., St. Louis, MO, USA) was utilized for EBRT, delivered by using a linear accelerator (Siemens, Erlangen, Germany) with 10- or 15-MV photon beams.
3. The SBRT techniqueContrast-enhanced CT with 2-mm slice thickness was performed for SBRT planning, with each patient in the supine position with both arms raised above the head. All patients were immobilized by using a customized vacuum cushion fixed in a stereotactic body frame (Smithers Medical Products Inc., North Canton, OH, USA). Diaphragmatic movements were reduced by using an abdominal compression device attached to the frame. SBRT dose planning and delivery were performed by using the On-Target planning system and CyberKnife (Accuray Inc., Sunnyvale, CA, USA) in 31 patients and the Eclipse treatment planning system and RapidArc (Varian Medical Systems Inc., Palo Alto, CA, USA) in 27 patients. The gross tumor volume (GTV) was defined as the visible lesion detected on CT. A thin-slice CT was performed with a slice thickness of 2 mm at 3 seconds per slice. These relatively slow CT images included respiratory movement of the target; therefore, the tumor volumes used during planning were larger than the GTVs and are referred to as internal target volumes (ITVs). The planning target volume (PTV) included an expansion of the ITV by 2-mm anteroposteriorly, 2-mm laterally, and 4-mm craniocaudally. Radiation doses were prescribed at 70%-80% isodose line of the maximum dose in CyberKnife and at 92%-99% in RapidArc to cover at least 95% of the PTVs.
Individualized prescribed doses, doses per fraction, and numbers of fractions were determined based on tumor size, location, patient performance, and organs at risk. Prescribed doses for SBRT only patients were 30-60 Gy (median, 45 Gy) in 3-5 fractions (median, 3 fractions). Prescribed doses for EBRT + SBRT patients were 15-18 Gy (median, 16 Gy) in 1 fraction. The SBRT doses were converted to the biologically equivalent dose at a fraction size of 2 Gy (BED2Gy) using a linear quadratic equation, BED = total dose × (1 + dose per fraction / α/β). The converted dose BED2Gy (α/β = 10) ranged from 48 to 150 Gy (median, 86 Gy).
4. Follow-up, response, and toxicity assessmentsTumor response was assessed as described in the Response Evaluation and Criteria in Solid Tumors (RECIST) v.1.1 at 3 months after SBRT completion. Local failure was defined as an increase in tumor size or the development of a new lesion in the radiation field. Distant failure was defined as the development of a new lesion in non-targeted liver or beyond the liver.
Acute and late toxicities were defined based on patient's symptoms, laboratory findings, and diagnostic findings that developed before or after 3 months post-SBRT, respectively. Toxicities were scored using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) v.4.03.
5. Statistical analysisSurvival and disease control were calculated from the start date of RT by using the Kaplan-Meier method. Various variables were evaluated from patient records for prognostic factor analysis. Log-rank analysis was used to compare prognostic factors. Those found to influence prognosis in univariate analysis were entered into multivariate analysis with the Cox proportional hazard regression model by using a backward conditional stepwise procedure to determine whether they acted independently. The Pearson chi-square test was used to compare incidences between two groups. All calculations were done with SPSS ver. 13.0 (SPSS Inc., Chicago, IL, USA), and p-values of <0.05 were considered significant.
Results1. Overall survival and prognostic factorsFollow-up durations ranged from 1 to 97 months (median, 10 months), and 50 patients (86%) had died at the time of the analysis. The 1-year and 2-year overall survival rates and median survival were 45%, 20%, and 10 months, respectively. Disease progression-free survival rates at 1-year and 2-year were 26% and 23%, respectively, with a median time to disease progression of 5 months. Local control rate at 1-year and 2-year were 85% and 72%, respectively (Fig. 1). In the primary cancer group, 1-year and 2-year overall survival rates and median survival were 29%, 11%, and 5 months, respectively. In the recurrent cancer group, 1-year and 2-year overall survival rates and median survival were 53%, 28%, and 13 months, respectively (Fig. 2). The statistically significant factors for local control and overall survival rates on univariate analysis are shown in Table 2. In the patients with small tumor (<50 mL) group, 1-year and 2-year overall survival rates and median survival were 61%, 29%, and 15 months, respectively. In the patients with large tumor (≥50 mL) group, 1-year and 2-year overall survival rates and median survival were 24%, 5%, and 5 months, respectively. Two groups showed statistically significant difference in overall survival rate (p = 0.004) (Fig. 3). In multivariate analysis, ECOG performance score (0-1 vs. 2-3; p = 0.037) and tumor volume (<50 vs. ≥50 mL; p = 0.030) were statistically significant factors for overall survival.
In the recurrent tumor group, univariate analysis showed that treatment volume (<50 vs. ≥50 mL) and time interval from surgery to recurrence (≤12 vs. >12 months) were significant factors for overall survival. The median survival of the patients with recurrence ≤12 months from the date of initial surgery was 8 months, whereas the median survival of those with recurrence >12 months was 17 months (p = 0.007). In multivariate analysis, time interval to recurrence was the only significant prognostic factor (p = 0.026) for overall survival.
2. ToxicitySeventeen patients (29%) experienced nausea, vomiting, abdominal pain, and systemic discomfort of grade 1 or 2 during treatment. During follow-up, 6 patients (10%) experienced a severe complication above grade 3. Of the 6 patients, three patients with grade 3 complication were managed by medication and recovered. Of the 3 patients with grade 4 complication, 2 patients with cholangitis and bile duct stenosis were hospitalized and successfully managed by medication and percutaneous transhepatic biliary drainage, respectively. One patient with gastric perforation was recommended to receive surgical treatment. However, she refused the surgery and expired. Details for toxicities are summarized in Table 3.
3. ResponseAmong the 58 patients, 54 patients were assessable for the radiological response 3 months after SBRT completion. Four out of 54 patients experienced a complete response, 18 patients a partial response, 27 patients a stable disease, and 5 patients experienced disease progression. The overall RECIST response rate was 38% (complete response 5%; partial response 31%) with a stable disease rate of 47%.
Discussion and ConclusionThe general approach for unresectable cholangiocarcinoma has been conventional EBRT with or without chemotherapy. Most of studies concerning EBRT are retrospective, nonrandomized, often combine gall bladder cancers with intrahepatic and extrahepatic cholangiocarcinoma, and involve small numbers of patients. In spite of limitations, they usually reported high rate of local recurrence (36%-72%) as a main treatment failure pattern with median survival of 9.5-15.8 months [4,7,8,9]. Various approaches including intraluminal brachytherapy have been made to increase local control rate. A study by Shin et al. [10] treated 31 patients with unresectable extrahepatic bile duct malignancies with EBRT alone (group 1) or EBRT + intraluminal brachytherapy (group 2). EBRT was delivered with a total dose ranging from 36 to 55 Gy (median, 50.4) in both groups. The prescribed dose of the brachytherapy for the group 2 was 15 Gy in 3 fractions. The median survival for overall group was 6 months (5 months for group 1 and 9 months for group 2). In the pattern of failure analysis, they report local failure rate as 58% (10 of 17 patients) for group 1 and 36% (5 of 14 patients) for group 2 [10]. A study by Alden and Mohiuddin [5] treated 48 patients with extrahepatic bile duct cancers. The median survival of those treated to >55 Gy was 24 months, vs. the median survival of those receiving <55 Gy was 6 months (p = 0.0003). A dose response is further suggested by the elongated median survival with increasing radiation dose (4.5, 9, 18, and 25 months for <45, 45-55, 55-65, and 66-70 Gy, respectively) [5]. Intraluminal brachytherapy seems to be a useful modality for increasing local control rate. However, it has several demerits of invasiveness and ambiguity in dose calculation. In this aspect, SBRT can be a promising modality of treatment for dose escalation for target and normal tissue saving. Especially in treating intrahepatic cholangiocarcinoma, SBRT may be a means to deliver precise therapeutic doses of radiation to the tumor while sparing the normal liver to avoid subsequent liver dysfunction. Several studies have shown SBRT results with few patients enrolled [3,11,12,13]. The largest series until now include 27 patients with unresectable cholangiocarcinoma treated with SBRT [12]. The dose used on all patients in this study was 45 Gy in 3 fractions, prescribed to isocenter. They note median progression-free and overall survivals of 6.7 and 10.6 months, respectively. We report median progression-free and overall survivals of 5 and 10 months. In their study, median CTV volume was 32 mL and tumor size ranged from 9 to 205 mL. Considering that the current study demonstrated tumor size as a prognostic factor for overall survival, our result is comparable to their result because we have treated more huge tumor ranging from 5 to 1,287 mL (median, 40 mL). In our study, although it is not statistically significant, patients with recurrent tumors show better results in local control and overall survival compared to patients with primary tumors. Dose prescribed to each group was not statistically different (BED2Gy 50-138 Gy, median BED2Gy 84 Gy for primary tumors versus BED2Gy 48-150 Gy, median BED2Gy 94 Gy for recurrent tumors, p = 0.439). However, tumor volume was significantly different between two groups (23-1,287 mL, median 143 mL for primary tumors versus 5-371 mL, median 18 mL for recurrent tumors, p < 0.001). According to this result, it seems that tumor size is more important prognostic factor for overall survival than prescription dose. In the recurrent tumor group, the interval from the initial surgery to recurrence has important prognostic significance. Several studies dealing with salvage treatment for recurrent cancer suggest that the interval from the initial treatment to recurrence is a significant prognostic factor for overall survival [14,15,16,17]. Based on this finding, we suppose that the interval from the initial treatment to recurrence would act as an important prognostic factor in this clinical setting. Therefore, we suggest that it would be beneficial to treat small and indolent recurrent tumors with SBRT as a salvage treatment.
In terms of complications, we report 10% (6 of 58 pts) of severe complications (Table 3). It is noteworthy that, of the 6 patients, 3 patients were re-irradiated patients (50%). The previously irradiated doses ranged from 45 to 60 Gy in conventional fractionation. If we exclude the re-irradiated patients, severe toxicity rate would be 5%. A special attention is required before giving re-irradiation.
This study suggests that SBRT can be considered as an effective local modality for unresectable primary or recurrent cholangiocarcinoma. Especially, patients with small and indolent recurrent cholangiocarcinoma are favorable candidates for salvage SBRT. Further, a well-controlled prospective study is necessary to confirm the optimal dose and constraints for SBRT in a large number of patients.
References1. Tan JC, Coburn NG, Baxter NN, Kiss A, Law CH. Surgical management of intrahepatic cholangiocarcinoma: a population-based study. Ann Surg Oncol 2008;15:600–608, PMID: 17987347.
2. Cunningham SC, Choti MA, Bellavance EC, Pawlik TM. Palliation of hepatic tumors. Surg Oncol 2007;16:277–291, PMID: 17935975.
3. Barney BM, Olivier KR, Miller RC, Haddock MG. Clinical outcomes and toxicity using stereotactic body radiotherapy (SBRT) for advanced cholangiocarcinoma. Radiat Oncol 2012;7:67PMID: 22553982.
4. Crane CH, Macdonald KO, Vauthey JN, et al. Limitations of conventional doses of chemoradiation for unresectable biliary cancer. Int J Radiat Oncol Biol Phys 2002;53:969–974, PMID: 12095564.
5. Alden ME, Mohiuddin M. The impact of radiation dose in combined external beam and intraluminal Ir-192 brachytherapy for bile duct cancer. Int J Radiat Oncol Biol Phys 1994;28:945–951, PMID: 8138448.
6. Deodato F, Clemente G, Mattiucci GC, et al. Chemoradiation and brachytherapy in biliary tract carcinoma: long-term results. Int J Radiat Oncol Biol Phys 2006;64:483–488, PMID: 16242254.
7. Ben-David MA, Griffith KA, Abu-Isa E, et al. External-beam radiotherapy for localized extrahepatic cholangiocarcinoma. Int J Radiat Oncol Biol Phys 2006;66:772–779, PMID: 17011452.
8. Ben-Josef E, Normolle D, Ensminger WD, et al. Phase II trial of high-dose conformal radiation therapy with concurrent hepatic artery floxuridine for unresectable intrahepatic malignancies. J Clin Oncol 2005;23:8739–8747, PMID: 16314634.
9. Chen YX, Zeng ZC, Tang ZY, et al. Determining the role of external beam radiotherapy in unresectable intrahepatic cholangiocarcinoma: a retrospective analysis of 84 patients. BMC Cancer 2010;10:492PMID: 20840777.
10. Shin HS, Seong J, Kim WC, et al. Combination of external beam irradiation and high-dose-rate intraluminal brachytherapy for inoperable carcinoma of the extrahepatic bile ducts. Int J Radiat Oncol Biol Phys 2003;57:105–112, PMID: 12909222.
11. Tse RV, Hawkins M, Lockwood G, et al. Phase I study of individualized stereotactic body radiotherapy for hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J Clin Oncol 2008;26:657–664, PMID: 18172187.
12. Kopek N, Holt MI, Hansen AT, Hoyer M. Stereotactic body radiotherapy for unresectable cholangiocarcinoma. Radiother Oncol 2010;94:47–52, PMID: 19963295.
13. Polistina FA, Guglielmi R, Baiocchi C, et al. Chemoradiation treatment with gemcitabine plus stereotactic body radiotherapy for unresectable, non-metastatic, locally advanced hilar cholangiocarcinoma. Results of a five year experience. Radiother Oncol 2011;99:120–123, PMID: 21621289.
14. Kim JS, Kim JS, Kim SY, Kim Ki, Cho MJ. Hyperfractionated radiotherapy with concurrent chemotherapy for para-aortic lymph node recurrence in carcinoma of the cervix. Int J Radiat Oncol Biol Phys 2003;55:1247–1253, PMID: 12654434.
15. Yahara K, Ohguri T, Imada H, Yamaguchi S, Terashima H, Korogi Y. Cancer of the pelvis: definitive three-dimensional conformal radiotherapy for patients with isolated recurrence in the para-aortic lymph nodes. J Radiat Res 2012;53:735–741, PMID: 22843374.
16. Maneo A, Landoni F, Cormio G, et al. Concurrent carboplatin/5-fluorouracil and radiotherapy for recurrent cervical carcinoma. Ann Oncol 1999;10:803–807, PMID: 10470427.
17. Sommers GM, Grigsby PW, Perez CA, et al. Outcome of recurrent cervical carcinoma following definitive irradiation. Gynecol Oncol 1989;35:150–155, PMID: 2807004.
Fig. 1Kaplan-Meier survival analysis for overall survival (OS), disease progression-free survival (DPFS), and local control (LC) rates for all patients (n = 58). SBRT, stereotactic body radiotherapy. 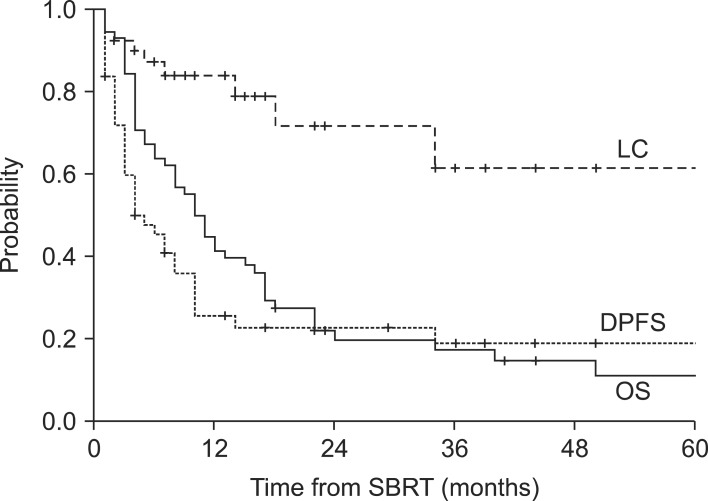
Fig. 2Kaplan-Meier overall survival (OS) graph for patients with primary (n = 28) versus recurrent (n = 30) cholangiocarcinoma. SBRT, stereotactic body radiotherapy. 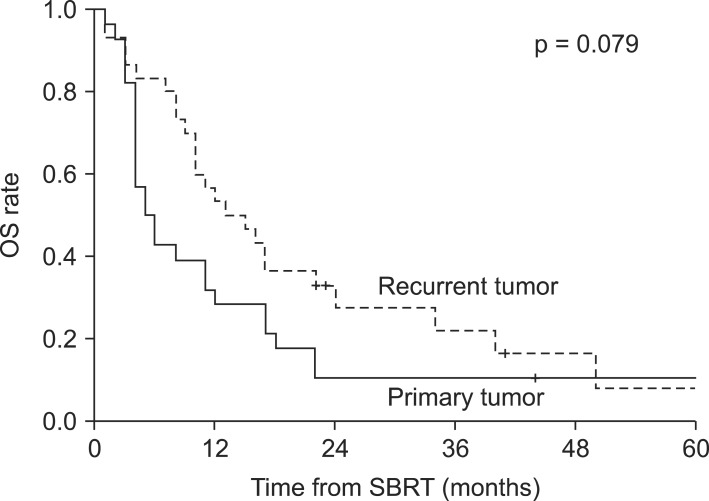
Fig. 3Kaplan-Meier overall survival (OS) difference in patients with small (<50 mL) tumor and large (≥50 mL) tumor. SBRT, stereotactic body radiotherapy. 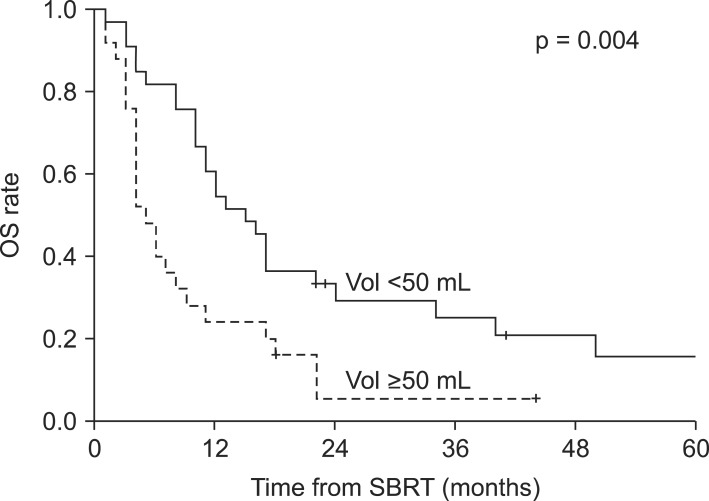
Table 2Prognostic factors affecting LC and OS rates on univariate analysis 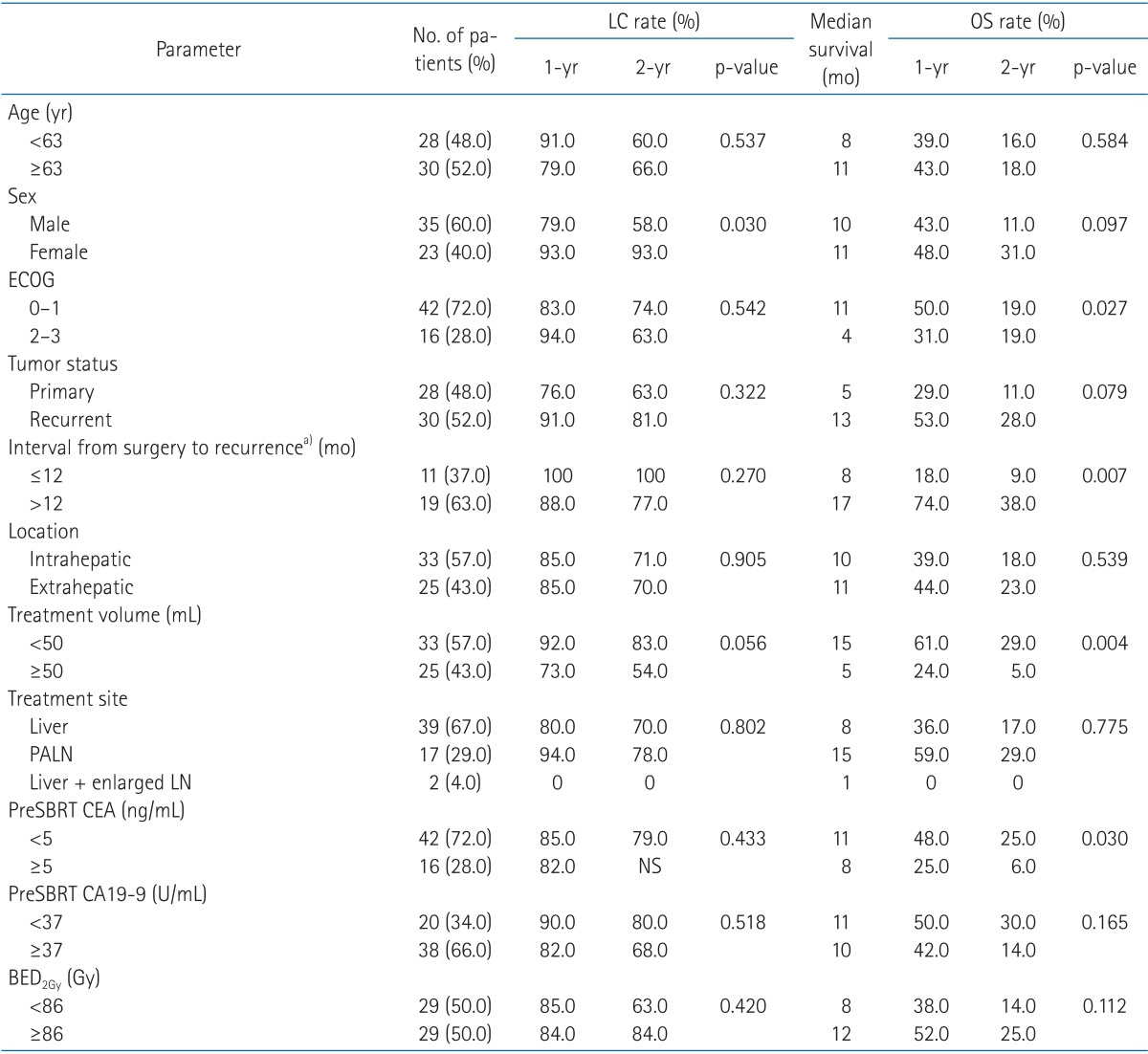 LC, local control; OS, overall survival; ECOG, Eastern Cooperative Oncology Group; SBRT, stereotactic body radiotherapy; CEA, carcinoembryonic antigen; CA19-9, carbohydrate antigen 19-9; BED2Gy, biologically equivalent dose in 2 Gy/fraction; NS, non-specific. a)Interval from surgery to recurrence includes recurrent tumor group only. |
|
||||||||||||||||||||||||||||||||||||||||||||||||

 |
 |