 |
 |
AbstractPurposeAn optimal once-daily radiotherapy (RT) regimen is under investigation for definitive concurrent chemoradiotherapy (CCRT) in limited disease small cell lung cancer (LD-SCLC). We compared the efficacy and safety of dose escalation with intensity-modulated radiotherapy (IMRT).
Materials and MethodsBetween January 2016 and March 2021, patients treated with definitive CCRT for LD-SCLC with IMRT were retrospectively reviewed. Patients who received a total dose <50 Gy or those with a history of thoracic RT or surgery were excluded. The patients were divided into two groups (standard and dose-escalated) based on the total biologically effective dose (BED, α/β = 10) of 70 Gy. The chemotherapeutic regimen comprised four cycles of etoposide and cisplatin.
ResultsOne hundred and twenty-two patients were analyzed and the median follow-up was 27.8 months (range, 4.4 to 76.9 months). The median age of the patients was 63 years (range, 35 to 78 years) and the majority had a history of smoking (86.0%). The 1- and 3-year overall survival rates of the escalated dose group were significantly higher than those of the standard group (93.5% and 50.5% vs. 76.7% and 33.3%, respectively; p = 0.008), as were the 1- and 3-year freedom from in-field failure rates (91.4% and 66.5% vs. 73.8% and 46.9%, respectively; p = 0.018). The incidence of grade 2 or higher acute and late pneumonitis was not significantly different between the two groups (p = 0.062, 0.185).
IntroductionSmall cell lung cancer (SCLC) is a rapidly progressive cancer if not controlled with appropriate intervention [1]. Only one-third of patients with SCLC are diagnosed with limited disease that can be encompassed within the radiotherapy (RT) field [2]. Given that SCLC is a relatively radiosensitive tumor [3] and the number of surgical candidates for very early-stage SCLC is small, RT is the main local treatment modality. Based on the Japan Clinical Oncology Group study 9104 [4], National Cancer Institute of Canada Clinical Trials Group data [5], and Yugoslavia data [6], concurrent chemoradiotherapy (CCRT) is the standard of care for limited disease small cell lung cancer (LD-SCLC) to improve survival.
The prescribed RT dose for SCLC has traditionally been lower than that for non-small-cell lung cancer due to the difference in radiosensitivity between the two tumor types [3]. However, recent studies have trialed modified schedule or escalated-dose RT to overcome locoregional recurrences after conventional-fractionated CCRT. The Intergroup 0096 study demonstrated that patients treated with a 45 Gy/30 fractions twice-daily schedule showed longer overall survival (OS) compared to those treated with 45 Gy/25 fractions once-daily [7], while the CONVERT [8] and CALGB 30610 [9] trials showed a similar OS in the 45 Gy/30 fractions twice-daily group compared to the conventional RT group. Despite the accelerated RT schedule showing improved survival [7], it also increased treatment-related toxicities and deteriorated the patients’ quality of life [7,8,10].
Conventional RT remains the standard scheme for LD-SCLC in many institutions [11] because two hospital visits per day tends to reduce patient compliance. Therefore, in our institution, we escalated the total dose but maintained a single daily dose of curative intent CCRT in patients with LD-SCLC. We analyzed the clinical outcomes of the patients with the aim to prove the efficacy of dose escalation.
Materials and Methods1. Patients (study population)We retrospectively reviewed patients with LD-SCLC who were treated with RT from January 2016 to March 2021 at our institution. Patients who were initially diagnosed, pathologically confirmed, treated with definitive aim CCRT with intensity-modulated radiotherapy (IMRT), and irradiated with a total dose of ≥50 Gy were included. Patients who had no follow-up chest computed tomography (CT) after treatment, a history of other malignancy in the 2 years prior to SCLC diagnosis, and experience of RT or surgery in the thoracic cavity were excluded (Fig. 1). This study was approved by the Institutional Review Board of Asan Medical Center (IRB No. 2023-0815). Informed consent was waived based on the retrospective nature of this study.
2. Treatment1) RadiotherapyFour-dimensional CT simulation was performed as the standard protocol, and the gross tumor volume (GTV) was delineated in the most expiratory phase. For patients who underwent induction chemotherapy, post-chemotherapy tumors, including all initial tumor sites, were delineated as the GTV. The clinical target volume (CTV) was delineated to cover the adjacent nodal area or ambiguous tumor margin in 48 patients (39.3%). The internal target volume (ITV) was added to the GTV or CTV to allow for respiratory tumor motion. The planning target volume (PTV) was expanded 3–7 mm from the ITV. All treatment was planned using IMRT with a total dose in the range of 50–66 Gy and conventional fractionation. Thereafter, the biological equivalent dose (BED) was calculated using the linear quadratic formula [12] to equally compare different treatments with different radiation doses per session using an α/β of 10. To evaluate the radiation dose irradiated to normal organs, the maximum dose, mean dose irradiated to each organ and Vx were measured, where Vx refers to the volume of the area irradiated more than x Gy. All patients were treated with image-guided radiotherapy (IGRT) with cone-beam CT conducted at least once a week. Using IGRT, the early response of tumor could be checked during treatment period, which allowed for adjustments to the treatment plan to reduce the treatment volume, as well as the normal organ radiation dose (e.g., lungs, esophagus, and heart).
2) ChemotherapyThe standard chemotherapy regimen included four cycles of platinum-based combination treatment as follows: etoposide 100 mg/m2/day over 3 hours on days 1–3, cisplatin 70 mg/m2 over 1 hour on day 1, and carboplatin at an area under the concentration-time curve of 5 over 1 hour on day 1. Chemotherapy was administered after the evaluation of hematologic toxicity and performance status by clinicians every 3 weeks. Two patients switched etoposide with irinotecan due to toxicity (irinotecan 65 mg/m2/day over 90 minutes on days 1 and 8). A total of 103 patients (84.4%) were administered 1–2 cycles of induction chemotherapy before CCRT due to old age, bulky tumor, or prompt start of treatment.
3. Evaluation after treatment and toxicitiesFollowing completion of CCRT, chest CT was performed 1 month later, followed by every 3 months for the evaluation of treatment response and surveillance of disease progression for the first 2 years, and every 6 months until 5 years. The maximum treatment response after definitive CCRT was decided by clinicians with the aid of chest CT and fluorodeoxyglucose positron emission tomography-CT if deemed necessary. In cases without radiation-induced pneumonitis, a complete response was defined as disappearance of the primary lesion or no obvious metabolic hyperactivity. A partial response required ≥30% decrease in the sum of the longest diameters (SLD) of target lesions. Between complete response and partial response, near complete response was considered in cases with ambiguous chest CT imaging; for example, radiation pneumonitis obscuring the total primary lesion with a minimal residual solid part with unclear metabolic activity. A progressive disease required ≥20% increase in the SLD and stable disease was defined as being neither partial response nor progressive disease.
Brain magnetic resonance imaging was performed at the time of initial diagnosis. Prophylactic cranial irradiation (PCI) was considered for patients without brain metastasis and with a partial or complete disease response after thoracic CCRT. PCI was selectively administered according to age, performance, and patient preference.
Toxicity was graded according to the Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. [13] After the completion of RT, RT-associated toxicity was graded as acute when it occurred within 3 months, and as late when it occurred after 3 months.
4. Statistical analysisLocoregional failure (LRF) was defined as tumor recurrence in the ipsilateral hemithorax and regional lymph node(s) and was subdivided into in-RT-field failure (IFF) and out-RT-field failure (OFF). Between the date of IFF and OFF, the LRF date was recorded as the earlier. Freedom from IFF (FFIFF), freedom from OFF (FFOFF), freedom from LRF (FFLRF), and freedom from distant metastasis (FFDM) were calculated from the date of diagnosis to the date of failure or the last follow-up (the date of the most recent cancer-associated clinical visit).
The disease progression date was recorded as the earliest date of any recurrence or the date of death in patients without recurrence. The survival status of the patients and the date of death were obtained through the national health insurance database. For the patients who died, the date of death was obtained and for the patients who remained alive, May 31, 2023, was recorded as the date of death. Additionally, progression-free survival (PFS) and OS were calculated from the date of diagnosis to the date of disease progression and death, respectively.
The Pearson chi-square test and independent sample t-test were used to compare the characteristics of patients in both groups. The Kaplan–Meier method was used to analyze the survival outcomes, and log-rank tests were used to evaluate the statistical significance between two groups. Clinical and therapeutic factors that may affect IFF and OS were analyzed, including factors that were significantly different between the two groups, using the Cox proportional hazard model in univariate analysis. Thereafter, multivariate analysis was performed using the backward elimination method in the Cox proportional hazard model including the factors with p-value <0.1 in univariate analysis or clinicians thought might be affecting each survival outcomes. Decided at clinician’s discretion, the overall stage was additionally included in the FFIFF analysis, and overall stage and whether PCI was performed were included in the OS analysis. A p-value <0.05 was considered statistically significant. SPSS version 21 (IBM, Armonk, NY, USA) was used for all statistical analyses.
Results1. Patient and disease characteristicsA total of 122 patients were analyzed, with a median follow-up period of 27.8 months (range, 4.4 to 76.9) (Table 1). The median age of the patients was 63 years (range, 35 to 78 years) and the majority of patients were male (104 patients, 85.2%), and had a history of smoking (105 patients, 86.0%). A total of 109 patients (89.3%) had a primary tumor of ≤7 cm and 114 patients (93.4%) had nodal metastasis. Only two patients had stage I disease according to the 8th staging system of American Joint Committee on Cancer (AJCC) [14] and all the patients had LD according to the International Association for the Study of Lung Cancer recommendation [15].
The patients were divided into a standard group, irradiated with a total dose of <BED10 70 Gy (median 63.5 Gy; range, 60.0 to 68.6 Gy) and a dose-escalated group, irradiated with a total dose of ≥BED10 70 Gy (median 73.7 Gy; range, 71.1 to 80.5 Gy). In the standard group, 53 patients (88.3%) had stage III compared to 40 patients (64.5%) in the dose-escalated group (p = 0.005). Additionally, the standard group showed a tendency toward higher nodal stage (p = 0.053), but there was no significant difference in forced vital capacity, forced expiratory volume in one second, and underlying interstitial lung disease or chronic obstructive pulmonary disease between the two groups. The diffusing capacity of the lung for carbon monoxide (DLCO) was better in the dose-escalated group (p = 0.011).
A total of 103 patients (84.4%) were treated with induction chemotherapy and 45 patients (36.9%) underwent PCI after definitive CCRT for thoracic disease (Table 2). Forty-eight patients (39.3%) had CTV for thoracic RT, including 25 (41.7%) in the standard group and 23 (37.1%) in the dose-escalated group. The median PTV was 292.2 mL (range, 39.5 to 742.3 mL) and 190.8 mL (range, 77.7 to 801.0 mL) in the standard and dose-escalated groups, respectively (p = 0.005).
2. Treatment outcomesThe median OS was 23.1 months (95% confidence interval [CI], 17.2–29.0) months and 38.7 months (95% CI, 28.4–49.0) in the standard and dose-escalated groups, respectively, with 1- and 3-year OS rates of 76.7% and 33.3% and 93.5% and 50.5%, respectively (p = 0.008) (Fig. 2). Fig. 3A–3E shows the Kaplan–Meier method survival curves of FFLRF, FFIFF, FFOFF, FFDM, and PFS, respectively. The median FFIFF was unreached in the dose-escalated group, but showed a significant difference compared to that of the standard group at 1 and 3 years (standard group 73.8% and 46.9% vs. dose-escalated group 91.4% and 66.5%, respectively; p = 0.018). Moreover, the 3-year FFLRF was 40.5% and 52.8% in the standard and dose-escalated groups (p = 0.028), while the 3-year FFDM was 28.4% and 50.6%, respectively (p = 0.034). None of the patients showed disease progression immediately after CCRT, and objective response ratio was 97.5% (n = 119) (Table 3).
3. Initial patterns of failureFor isolated failure in-RT field, there were eight patients (13.3%) in the standard group and one patient (1.6%) in the dose-escalated group (p = 0.025) (Table 4). Sixteen patients (26.7%) did not experience recurrence in the standard dose group compared to 23 patients (37.1%) in the dose-escalated group showing a tendency for recurrence to be less in the dose-escalated group (p = 0.086). The initial failure site contained the RT field in 19 patients (31.7%) of the standard group and 11 patients (17.7%) of the dose-escalated group. The majority of them were found in combination with out-of-RT field recurrence or distant metastasis. The most frequent failure pattern contained distant metastasis in both groups.
4. Acute and late toxicitiesAmong all patients, 26 (21.3%) showed grade 2 or higher acute radiation pneumonitis and 28 (23.0%) showed grade 2 or higher acute radiation-induced esophagitis. Grade 2 or higher acute esophagitis occurred in 19 patients (31.7%) in the standard group and nine patients (14.5%) in the dose-increase group, showing a statistically significant difference between the two groups (p = 0.024) (Table 5). Grade 2 or higher late radiation pneumonitis or fibrosis was identified in 19 patients (15.6%) of all patients and only one patient suffered from grade 2 late esophagitis. None of the patients presented with grade 4 or 5 toxicity. Additionally, V5, V10, V20 of the lungs and mean dose of the heart were lower in the dose escalated group with statistically significant difference (Supplementary Table S1).
5. Prognostic factorsIn univariate analysis for FFIFF, DLCO, CCRT regimen including cisplatin, and BED10 ≥70 Gy showed a significant favorable correlation. But in multivariate analysis only DLCO, and the CCRT regimen showed statistical significance with a hazard ratio (HR) of 0.980 (95% CI, 0.962–0.999; p = 0.036) and 0.502 (95% CI, 0.270–0.932; p = 0.029), respectively (Table 6). BED10 ≥70 Gy showed favorable tendency but was not statistically significant (HR=0.566; 95% CI, 0.311–1.028; p = 0.062). For OS, in multivariate analysis, the CCRT regimen remained statistically significant, with a HR of 0.498 (95% CI, 0.314–0.790; p = 0.003) and also BED10 ≥70 Gy showed a significant favorable correlation with a HR of 0.620 (95% CI, 0.392–0.981; p = 0.041) (Table 7).
Discussion and ConclusionOur study showed improved local control in the RT field, with a 1-year FFIFF of 91.4% in the dose-escalated group compared to the standard group. Additionally, for FFIFF, BED10 ≥70 Gy showed a favorable correlation in univariate analysis and was marginally significant in multivariate analysis. Furthermore, the dose-escalated group showed improved OS, with a median OS of 23.1 months and 38.7 months in the standard and dose-escalated groups, respectively. Therefore, we consider the possibility that improvement in local control led to improved OS. Even with total dose escalation for treatment volume, the dose-volumetric histogram parameters to normal organs could be maintained at a similar or even lower level of those of the standard group using IMRT, with the significantly lower incidence of grade 2 or higher acute esophagitis (p = 0.024).
Several retrospective and prospective studies have compared 45 Gy/30 fractions twice-daily with various once-daily regimens with a total dose of 45–70 Gy [7-10,16,17]. Although comparing survival rates between studies warrants caution, the median OS of the dose-escalated group in our study was not inferior to that of 45 Gy/30 fractions twice-daily groups in other studies, ranging from 23.0 to 31.4 months [7-10,16,17]. In the CONVERT [8] trial in 2017, the median OS of the 45 Gy twice-daily group was 30 months compared to 25 months in the 66 Gy once-daily group (p = 0.14). Moreover, the CALGB 30610 [9] trial presented in 2023 reported a median OS of 28.5 months and 30.1 months in the 45 Gy twice-daily group and 70 Gy once-daily groups, respectively (p = 0.498).
However, few studies have compared the once-daily regimen [18,19] for dose escalation. Kim et al. [18] showed a 3-year OS of 53.6% in the dose-escalated group irradiated with >54 Gy, and Tomita et al. [19] showed a median OS equal to 41.0 months in the standard fractionation group (≥54 Gy). In the current study, we reveal that the use of a once-daily regimen with IMRT achieves a median OS of more than 3 years, with acceptable toxicity in all patients. To the best of our knowledge, this study is the first to use IMRT in all patients and represents an important basis to clarify the optimal once-daily regimen with a modern RT technique.
The PTV were significantly larger in the standard group compared to those in the dose-escalation group (p = 0.005), but there was no significant difference in the GTV (p = 0.316), indicating no significant difference in the total primary and nodal gross tumor volumes. However, the overall TNM stage according to the 8th edition of AJCC [17] was significantly higher (p = 0.005) in the standard group, which may impact disease control or OS. Although most studies and guidelines divide SCLC into limited or extensive stages [20,21], several studies have announced the prognostic value of TNM staging in patients with SCLC [22,23]. To account for this, we performed additional multivariate analysis, including factors related to tumor extent, and found no statistically significant factors related to FFIFF and OS. However, as this is a limitation of our retrospective study, long-term follow-up with more patients and further randomized controlled studies considering patients and disease characteristics are needed.
The criterion for dividing the two groups in this study was BED10 70 Gy, and in terms of the RT regimen, its total dose reaches approximately 60 Gy in 2 Gy per fraction. A phase I study presented in 1998 sought to investigate the maximum tolerative dose (MTD) in SCLC starting with the fourth cycle of chemotherapy. The authors reported 70 Gy/35 fractions (BED10 80 Gy) as the MTD, which led to grade 4 or more acute esophagitis in 33% of patients, with no cases of acute pneumonitis [24]. Moreover, by implementing concurrent chemotherapy and conformal RT, the CONVERT [8] and CALGB 30610 [9] trials reported 19% and 17.5% of grade 2 or more acute esophagitis with 66 Gy/33 fractions and 70 Gy/35 fractions, respectively. Although prospective studies have introduced high doses [8,9,25], in retrospective studies [10,16,17], most of the institutions have only attempted modest dose escalations, increasing gradually from 50 Gy or more in conventional RT. In our institution, we employ dose escalation in selective patients with consideration for their age, performance, other morbidities, and dose constraints for normal organs. Among the few studies that have compared once-daily regimens, one showed no significant difference in complications based on 54 Gy [26] and another study showed better PFS and OS based on the same dose [18]. Therefore, we set a dose that was slightly higher than this as the standard and converted it into BED10 considering the diversity of the dose per fraction.
This study has a few limitations that warrant discussion, largely owing to its retrospective nature. First, some information could not be obtained, even after thoroughly reviewing the patients’ medical records, because some patients had participated in clinical phase III double-blind trials for which we could not establish whether the patients had received the systemic agent or not. However, only a small number of the included patients participated in such trials. Second, regarding treatment toxicity, data for grade 1 toxicity may have been missed when it was based on the patient’s subjective symptoms. However, as CTCAE version 5.0 defines grade 2 or higher toxicity as that requiring medication, toxicity of grade 2 or higher could be investigated without omission by making inquiries in relation to the prescribing history, which may be clinically important. Finally, the patient and disease characteristics, such as DLCO, overall stage, PTV and chemotherapy regimen may have varied between the two groups. Unlike the previous three factors, chemotherapy regimen was found to have significant effect on the survival rate in univariate and multivariate analyses. However, as we had no strict criteria on chemotherapy such as dose and time intervals, and also included a small number of double-blind trials of systemic therapy, the influence of additional confounders cannot be ruled out when interpreting results related to chemotherapy. Therefore, a well-designed randomized trial that can compensate for these group differences should be conducted.
Like the standard dose group, the most common failure pattern in the dose-escalated group included distant metastases. Therefore, the need for systemic therapies that can control distant metastases more efficiently and exhibit fewer side effects will continue to be important to extend the survival period. Several ongoing trials have introduced various drugs concurrently (NCT04602533, NCT04624204, NCT03811002) or as consolidation (NCT03540420, NCT03703297) [27]. If the standard regimen of systemic therapy changes according to the results of these trials, additional studies on the optimal RT regimen and the interaction between systemic therapy and RT will be needed.
In conclusion, dose-escalated once-daily CCRT with an IMRT technique over 70 Gy in BED10 affordably improved survival and local tumor control without increasing toxicity. A prospective study will be designed to overcome the limitations of these retrospective outcomes.
NotesStatement of Ethics This study was reviewed and approved by the Institutional Review Board at Asan Medical Center (IRB No. S2023-1355-0001). Author Contributions Conceptualization, Choi W, Song SY; Investigation and methodology: Yang E, Joo JH, Song SY; Writing of the original draft: Yang E, Song SY; Writing of the review and editing: Yang E, Shin YS, Joo JH, Choi W, Kim SS, Choi EK, Song SY; Formal analysis: Yang E, Shin YS; Data curation: Yang E, Lee J, Song SY. Supplementary MaterialsSupplementary materials can be found via https://doi.org/10.3857/roj.2023.00591.
Fig. 1.Patient flow chart. LD-SCLC, limited disease small cell lung cancer; CCRT, concurrent chemoradiation therapy; RT, radiotherapy; IMRT, intensity-modulated radiotherapy. 
Fig. 3.Other survival outcomes: (A) freedom from locoregional failure, (B) freedom from in-field failure, (C) freedom from out-of-field failure, (D) freedom from distant metastasis, (E) progression-free survival. BED, biologically equivalent dose. 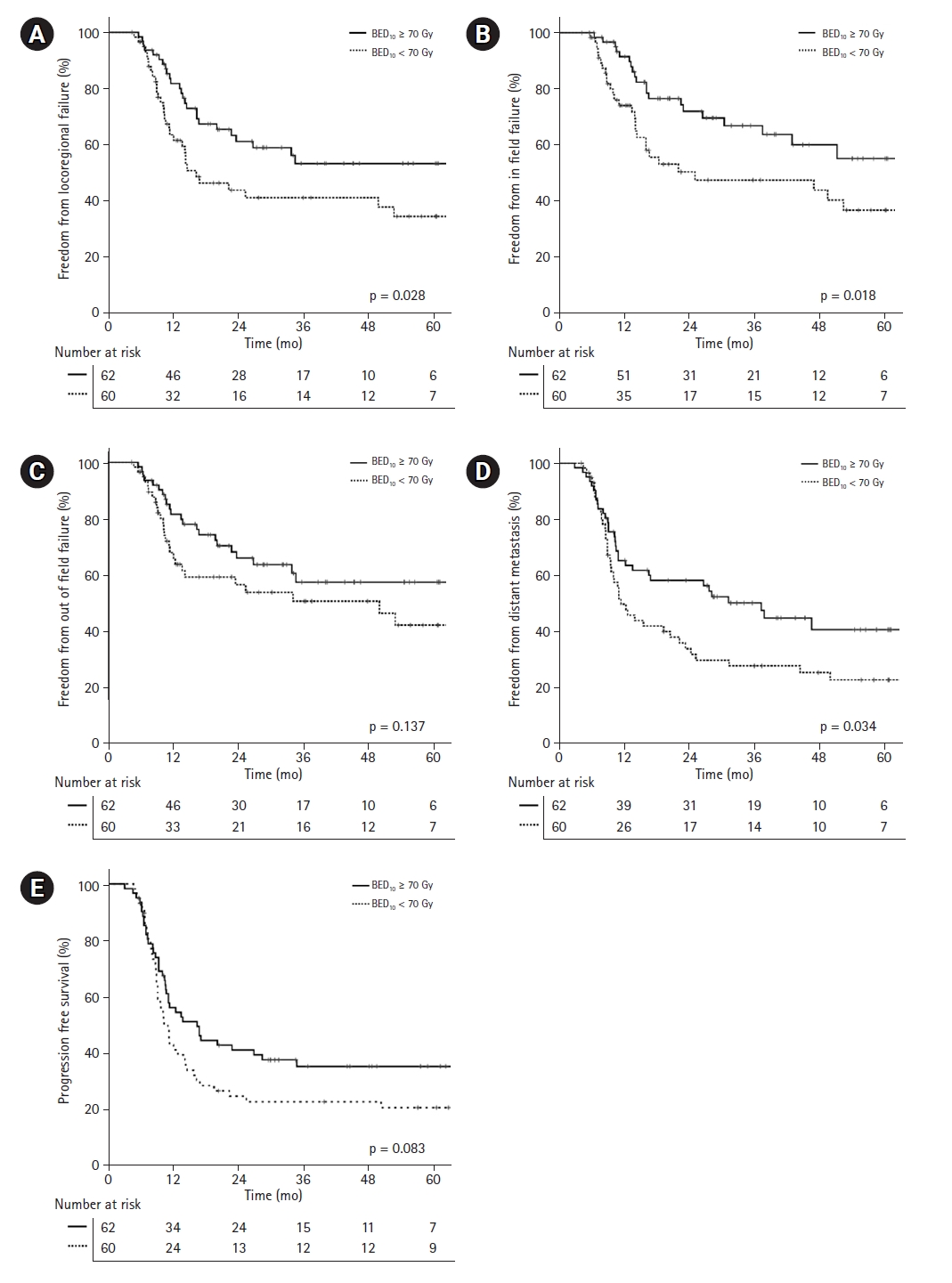
Table 1.Patient characteristics Table 2.Treatment Table 3.Treatment response Table 4.Initial patterns of failure according to the RT field Table 5.Acute and late toxicities using CTCAE v5.0 Table 6.Univariate and multivariate analysis for freedom from in-field failure Table 7.Univariate and Multivariate Analysis for Overall Survival References1. Domine M, Moran T, Isla D, et al. SEOM clinical guidelines for the treatment of small-cell lung cancer (SCLC) (2019). Clin Transl Oncol 2020;22:245–55.
3. Carmichael J, Degraff WG, Gamson J, et al. Radiation sensitivity of human lung cancer cell lines. Eur J Cancer Clin Oncol 1989;25:527–34.
4. Takada M, Fukuoka M, Kawahara M, et al. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with cisplatin and etoposide for limited-stage small-cell lung cancer: results of the Japan Clinical Oncology Group Study 9104. J Clin Oncol 2002;20:3054–60.
5. Murray N, Coy P, Pater JL, et al. Importance of timing for thoracic irradiation in the combined modality treatment of limited-stage small-cell lung cancer. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 1993;11:336–44.
6. Jeremic B, Shibamoto Y, Acimovic L, Milisavljevic S. Initial versus delayed accelerated hyperfractionated radiation therapy and concurrent chemotherapy in limited small-cell lung cancer: a randomized study. J Clin Oncol 1997;15:893–900.
7. Turrisi AT 3rd, Kim K, Blum R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med 1999;340:265–71.
8. Faivre-Finn C, Snee M, Ashcroft L, et al. Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol 2017;18:1116–25.
9. Bogart J, Wang X, Masters G, et al. High-dose once-daily thoracic radiotherapy in limited-stage small-cell lung cancer: CALGB 30610 (Alliance)/RTOG 0538. J Clin Oncol 2023;41:2394–402.
10. Han D, Hao S, Tao C, et al. Comparison of once daily radiotherapy to 60 Gy and twice daily radiotherapy to 45 Gy for limited stage small-cell lung cancer. Thorac Cancer 2015;6:643–8.
11. Farrell MJ, Yahya JB, Degnin C, et al. Radiation dose and fractionation for limited-stage small-cell lung cancer: survey of US radiation oncologists on practice patterns. Clin Lung Cancer 2019;20:13–9.
12. Barendsen GW. Dose fractionation, dose rate and iso-effect relationships for normal tissue responses. Int J Radiat Oncol Biol Phys 1982;8:1981–97.
13. US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0 [Internet]. Washington, DC: US Department of Health and Human Services; 2017 [cited 2023 Sep 13]. Available from; http://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf.
14. Amin MB, Edge SB, Greene FL, Byrd DR, Brookland RK, Washington MK, et al. AJCC cancer staging manual. 8th ed. Cham, Switzerland: Springer; 2017, p. 431–56.
15. Nicholson AG, Chansky K, Crowley J, et al. The International Association for the Study of Lung Cancer Lung Cancer Staging Project: Proposals for the Revision of the Clinical and Pathologic Staging of Small Cell Lung Cancer in the Forthcoming Eighth Edition of the TNM Classification for Lung Cancer. J Thorac Oncol 2016;11:300–11.
16. Watkins JM, Fortney JA, Wahlquist AE, et al. Once-daily radiotherapy to > or =59.4 Gy versus twice-daily radiotherapy to > or =45.0 Gy with concurrent chemotherapy for limited-stage small-cell lung cancer: a comparative analysis of toxicities and outcomes. Jpn J Radiol 2010;28:340–8.
17. Gazula A, Baldini EH, Chen A, Kozono D. Comparison of once and twice daily radiotherapy for limited stage small-cell lung cancer. Lung 2014;192:151–8.
18. Kim BH, Chung JH, Son J, Kim S, Wu HG, Kim HJ. Analysis of once-daily thoracic radiotherapy dose according to the underlying lung disease in patients with limited-stage small cell lung cancer undergoing concurrent chemoradiotherapy. Cancer Res Treat 2023;55:73–82.
19. Tomita N, Kodaira T, Hida T, et al. The impact of radiation dose and fractionation on outcomes for limited-stage small-cell lung cancer. Int J Radiat Oncol Biol Phys 2010;76:1121–6.
20. Dingemans AC, Früh M, Ardizzoni A, et al. Small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2021;32:839–53.
21. Ganti AKP, Loo BW, Bassetti M, et al. Small cell lung cancer, version 2.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 2021;19:1441–64.
22. Shirasawa M, Fukui T, Kusuhara S, et al. Prognostic significance of the 8th edition of the TNM classification for patients with extensive disease small cell lung cancer. Cancer Manag Res 2018;10:6039–47.
23. Li J, Zhao Y, Li C, Zhu L, Liu C, Liu L. The revision of 8th edition TNM stage criteria is more accurate in prediction postoperative survival for SCLC patients. Int J Surg 2017;48:83–5.
24. Choi NC, Herndon JE 2nd, Rosenman J, et al. Phase I study to determine the maximum-tolerated dose of radiation in standard daily and hyperfractionated-accelerated twice-daily radiation schedules with concurrent chemotherapy for limited-stage small-cell lung cancer. J Clin Oncol 1998;16:3528–36.
25. Komaki R, Paulus R, Ettinger DS, et al. Phase II study of accelerated high-dose radiotherapy with concurrent chemotherapy for patients with limited small-cell lung cancer: Radiation Therapy Oncology Group protocol 0239. Int J Radiat Oncol Biol Phys 2012;83:e531–6.
|
|
|||||||||||||||||||||||||||||||||||||

 |
 |